R = Rydberg constant = 1.096776 × 10 7 m -1
Equation (ix) shows that frequency varies with the initial and the final orbital quantum number.
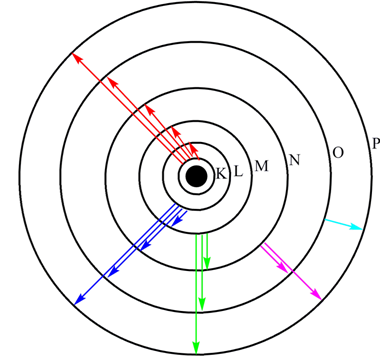
Figure 1.4. Bohr orbit of hydrogen and various series or spectral line. Layman(rd), Balmer (blue), Paschen (green), Brackett (magna), Pfund (cyno).
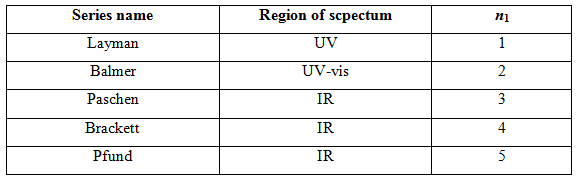
Table 1.2.
Merits and Demerits of Bohr Theory:
Merits: (i) In this theory the stability of an atom has been explained by introducing angular momentum of electron.
(ii) The origin or discontinuous line spectrum by atom can be explained by this theory.
Demerits: (i) In this model the concept of integer ( n ) has been introduced in selecting stable orbits for electrons. This was merely to fit the experimental data. There is no theoretical justification for it.
(ii) Employing instruments of high resolving power, single line in the spectrum of atomic hydrogen can also be resolved in several fine lines, “fine structure”. This phenomenon cannot be explained by Bohr´s theory.