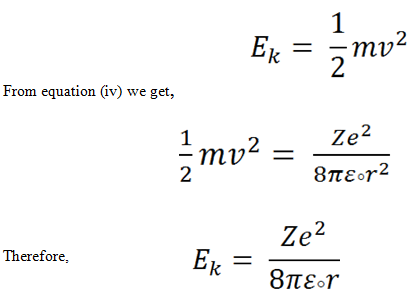Applying Bohr angular momentum we get,
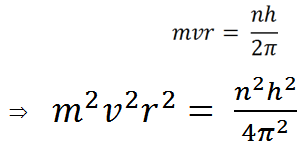 |
(iii) |
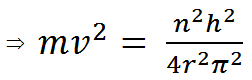 |
(iv) |
Therefore we can write,
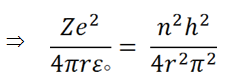 |
(v) |
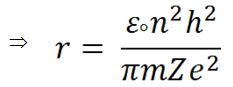 |
(vi) |
Note: r ∝ n2
Energy of an electron in a Bohr Orbit:
An electron in an atom possesses both kinetic energy that arises due to its motion around the nucleus and potential energy due to its attachment to the nucleus. Therefore, the total energy acquire by an electron will be the sum of both energies.
i . e . Total energy ( E t ) = Kinetic energy ( E k ) + Potential energy ( E p )
Kinetic energy:
Let us consider the mass of an electron is m , it circulates around the nucleus of charge Z along an orbit of radius r with a velocity of v .