Bohr´s model (1913):
Postulations:
(A) In a Rurtherford type atomic model, electrons can circulate around the positively charged nucleus only along certain selected orbits. During its circular movement along the elected orbit electron neither absorbs nor emits any radiation.
The angular momentum of the electron should be an integral multiple of h /2π ( h = Plank´s constant, 6.626 × 10-34 Js)
Let us consider an electron having mass m moving with a velocity of v along a orbit of radius r .
The angular momentum will be mvr .
According to Bohr´s theory,
mvr = nh /2π, n = any integer
1st orbit ( n = 1): mvr = 1 h /2π
2nd orbit ( n = 2): mvr = 2 h /2π
3rd orbit ( n = 3): mvr = 4 h /2π…………so on
Thu the angular momentum of an electron is quantized.
(B) When one electron jumps from one selected orbit to another, the emitted or absorbed energy should be equal to the energy difference in between two states as per quantum theory of radiation.
When an electron jumps from an orbit having energy E 1 to another orbit of energy E 2 (E 1>E 2 ), the frequency of the emitted radiation will be,
E 1 - E 2 = Δ E = h v
⇒ v = ( E 1 - E 2 )/ h = ΔE / h
This shows that discrete line will appear depending on ΔE value.
Radii of a Bohr Orbits:
In Bohr´s theory, the basic concept of Rutherford model has been maintained. The addition was the angular momentum.
Let us consider an electron of mass m , circulates around the nucleus of charge Z along an orbit of radius r with a velocity of v .
From Rutherford model we get that the centrifugal force will be equal to coulombic force,
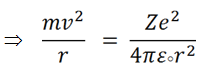 |
(i) |
ε0 = Permittivity of vacuum
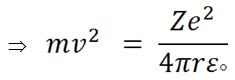 |
(ii) |