Potential energy:
Potential energy of an electron in an atom is given by the work to be done to bring an electron from infinity to its radius ( r ).
The total work done ( W ) will be then,
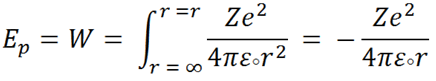 |
(vii) |
The negative sign implied that work has to be done to remove electron from the radius of the atom.
Total energy of the electron:
Total energy ( E t ) = Kinetic energy ( E k ) + Potential energy ( E p )
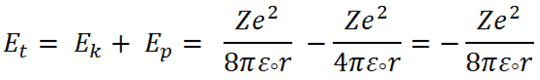 |
(viii) |
Interpretation of atomic spectra of hydrogen atom by Bohr´s Theory:
Let us consider two orbits having quantum number n 1 and n 2 , n 2 > n 1.
The energy corresponding to the orbits are, E n 1 and E n 2 , respectively.
Since, n 2 > n 1, E n 2 > E n 2
Hence, the energy difference ( Δ E ) will be equal to E n 2 - E n 2 .
(ix) |
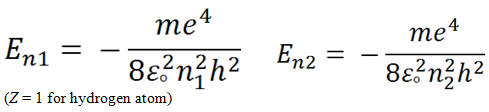
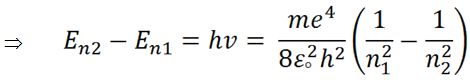 |
(x) |
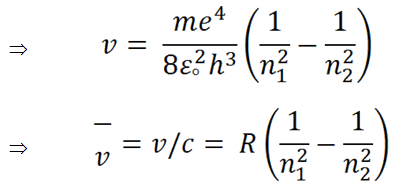 |
(xi) |