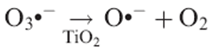Effect of O3
Adding ozone in dioxygen or air is a very efficient means of enhancing the photocatalytic rates of the removal and, above all, the mineralization of organic pollutants both in air and in water, even if the wavelengths are intentionally selected so as not to excite ozone.
This substantial effect is attributed to the difference in electron affinity between O3 (2.1 eV) and O2 (0.44 eV).
Consequently, in the presence of ozone, the electrons photopromoted to the TiO2 conduction band can be captured more easily, either directly:
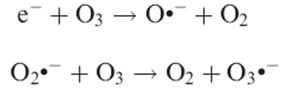
The radical anion O3._ is more unstable than O3 and can presumably split easily at the surface of TiO2: