Binding Energy (BE)
The Binding Energy (BE) is characteristic of the core electrons for each element. The BE is determined by the attraction of the electrons to the nucleus. If an electron with energy x is pulled away from the nucleus, the attraction between the electron and the nucleus decreases and the BE decreases. Eventually, there will be a point when the electron will be free of the nucleus.
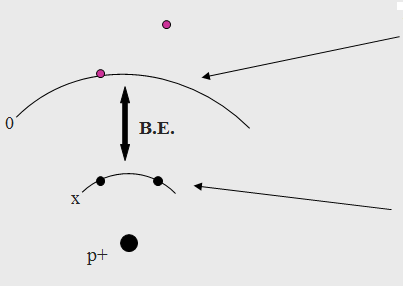 |
This is the point with 0 energy of attraction between the electron and the nucleus. At this point the electron is free from the atom. |
These electrons are attracted to the proton with certain binding energy x |