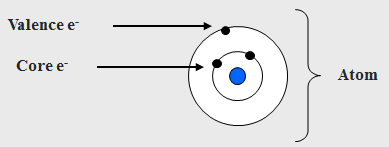
Why the Core Electrons?
• An electron near the Fermi level is far from the nucleus, moving in different directions all over the place, and will not carry information about any single atom.
• Fermi level is the highest energy level occupied by an electron in a neutral solid at absolute 0 temperature.
• Electron binding energy (BE) is calculated with respect to the Fermi level.
• The core e-s are local close to the nucleus and have binding energies characteristic of their particular element.
• The core e- s have a higher probability of matching the energies of AlKα and MgKα .