3.12.3.4. Cofactor Approaches
Metal-coordinated enzymes are everywhere in nature. A metallic species at the active site of an enzyme often plays a critical role in the reaction pathway by enhancing substrate selectivity and accelerating reaction rates and are the cofactor of the enzyme. Inspired by the efficiency of metalloenzymes, several efforts have been put forth toward the development of improved antibody catalysts with metals at the active site. Thus, antibody aldolase 38C2 was chosen as the parent antibody by several groups for the development of novel catalytic antibodies by the employment of cofactors. Nicholas et al. have employed bis-imidazolyl ligand coordinated copper complexes a cofactor which coordinate with LysH93 residue of 61 equipped with a reactive succinic anhydride moiety and CuCl2. This semisynthetic metalloantibody, 38C2-62-CuCl2, catalyzed the hydrolysis of picolic acid ester 62 in aqueous buffer under physiological conditions. This study exemplified that modification of the active site by a metal-coordinated ligand could alter the catalytic nature of the parent antibody, affording a catalyst with very different catalytic activity (Figure 3.44).
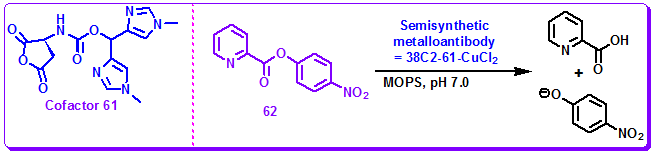
Figure 3.44: Cofactor 61 and the ester hydrolysis catalyzed by semisynthetic metalloantibody 38C2-61-CuCl2.
Pyridoxal 5′-phosphate (PLP, 63) has been shown to be an effective cofactor for antibody-catalyzed aldol and retro-aldol reactions. Aldolase antibody 10H2, elicited to hapten 64, and combined with cofactor PLP 63, catalyzed the aldol reaction between glycine and aldehyde 65, with a rate acceleration of double the background reaction where no PLP was applied (Figure 3.45a). This incorporation of PLP also improved the rates of the retro-aldol reactions of the threo- and erythro- isomers with rate enhancements of 4-fold and 2.5-fold, respectively (Figure 3.45b).
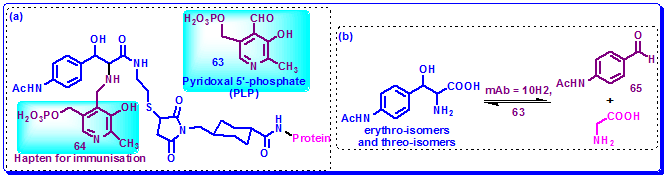
Figure 3.45: (a) Structure of PLP and hapten conjugated with carrier protein, (b) aldol and retro-aldol reactions catalyzed by mAb 10H2 with PLP 63.
Metalloporphyrins are well known in the research field of biological oxidation processes. Its application as a cofactor for a catalytic antibody was recently investigated. Thus, antibody SN37.4, elicited against a water soluble tin(IV) porphyrin 66 showed its oxidative activity upon assembly with a ruthenium(II) cofactor 67. This antibody-metalloporphyrin assembly showed enzymatic characteristic and transfer oxygen to substrate 68 with high enantioselective (Figure 3.46).
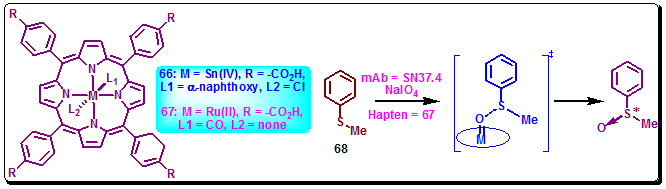
Figure 3.46: Metalloporphyrine haptens and the shown oxidation reaction catalyzed by antibody SN37.4.
3.12.3.5. Conclusion
From the above discussions and examples, it is clear that catalytic antibodies are efficient similar to the proficiency of natural enzymes in successful chemical transformation. Therefore a great attention has been drawn to this field of research. A large number of antibodies that are able to catalyze a variety of chemical processes have been investigated. Many advances based on the concept of catalytic antibodies as first described by Jencks, has been achieved. The ability to catalyse a chemical transformation stereospecifically and regiospecifically would offer the greatest immediate potential for their exploitation in the pharmaceutical and fine chemical industries for the synthesis and manufacture of new and purer drug candidates.
Antibodies that catalyze aminoacylation reaction could be used for facilitating aminoacylation of tRNAs with unnatural amino acids, an exciting focus towards expansion of genetic code and studies of protein biosynthesis. Efforts toward the exploitation of catalytic antibodies as therapeutics is the current research focus. It is also a hope that in near future potent catalytic antibody can be generated to treat diseases like HIV infection and Alzheimer’s disease, cancer. Phage-based screenings, selections and rational design can help one to engineer antibodies with anticancer activity and metallo-antibodies with hydrolytic activity.
Thus, these advancements, as well as future effort in this field of research will facilitate the discovery of more efficient catalytic antibodies that might provide synthetic chemists with novel tools to target challenging synthetic problems.
Clearly, the demonstration of the generality of catalytic antibodies and their role in understanding fundamental aspects of catalysis has generated excitement and opened new horizons of scientific and industrial opportunity that bridge enzymology, immunology and chemistry.