3.12.3.1.6. Antibody-Catalyzed Hydride Transfer Reaction
By raising antibodies to the appropriate hapten a variety of catalytic activities may be generated. For example, the compound in Figure 3.36has been synthesized to mimic a proposed combined NADH–NAD cofactor product transition state of hydride transfer in dehydrogenase enzymes. Thus compounds 24 and 25 were designed and prepared as stable TSAs for the hydride transfer process between 26 and an aliphatic aldehyde 28 (Figure 3.36). These haptens incorporated a rigid bicyclic structure containing a 3-piperidone oxime motif. The oxime mimics the carboxamide group in nicotinamide. The piperidone is held in the boat conformation corresponding to the transition state by a three-atom lactam bridge. The aldehyde carbon in the transition state was mimicked by a methylene group in Hapten 24. In hapten 25, the same is mimicked by a sulfonyl group. Thus, theses haptens were intended to use for the generation of catalytic antibodies with the potential to act as dehydrogenase mimics for hydride transfer reaction.
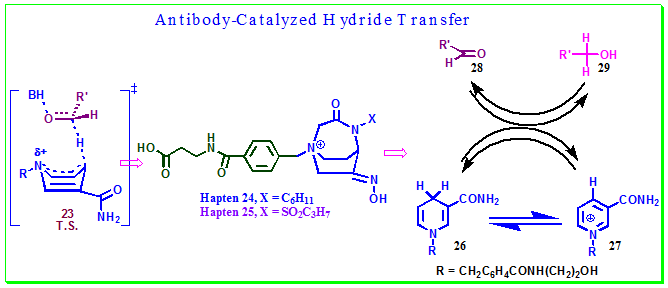
Figure 3.36: Hapten 24 and 25 were designed for the targeted hydride transfer reaction.
3.12.3.1.7. Disadvantages with TSA-Based Approaches
There are many reported instances wherein Antibody elicited against a TSA hapten is unable to catalyze the target reactions. This is due to the fact that the generation of an antibody (an immune response) upon immunization of a TSA hapten is driven by the affinity of the hapten rather than targeted catalytic activity of the produced immune response, the Antibody. Therefore, somatic mutations can raise antibodies that favor tighter hapten binding but are not suitable to perform catalysis.
Also many of the designed antibody catalysts are limited in their capability to accelerate chemical reactions as efficiently as natural enzymes in terms of efficiency [(kcat/KM)/Kuncat]. Published kcat/KM values of catalytic antibodies range from 102 to 104 M−1S−1, while those of natural enzymes range from 106 to 108 M−1S−1.
It is crucial that antibody catalysts raised against TSAs have to release the product to be considered efficient, Furthermore, in a number of cases it was found that because of high affinity of the antibody to bind the formed product tightly (which is again due of structural similarity of the product to the TSA haptens used in immunization), the products of the reaction can not be released. Therefore, the efficiency of the antibody is very low and can not be useful, which is a major case for many hydrolytic antibodies.
Another possibility for the poor performance of some transition state analog-based antibody catalysts may be an inability to design a stable organic compound that could reproduce the geometric criteria such as fractional bond orders, extended bond lengths, expanded valences, distorted bond angles, and charge distributions in the short-lived structure of a transition state.
The high catalytic proficiency of natural enzymes is due to the fact that the substrates remain bound buried in their catalytic machinery. However, in antibody catalysis, the moieties of the bound haptens that play a major role in catalytic activity and mimic the transition state are often positioned near the entrance of the antibody-combining site. This disparity in the overall architecture of natural enzymes and catalytic antibodies is undoubtedly a factor in the lower catalytic proficiency of the catalytic antibodies.
Therefore, antibody structure itself places limitations on the kind of reactions agreeable to catalysis. Thus, many other strategies have been developed to generate catalytic antibodies which act like enzyme as in general base and covalent catalysis, proximity effects, and the use of strain. We are going to exemplify few of such examples.
3.12.3.2. ‘Bait-and-switch’ Strategy
Based on the fact that a charged small molecule can mimic a charged amino acid residues in the antibody-binding site, a new approach for hapten design has been developed to expand the scope of antibody catalysis. In this strategy a point charge is placed on the hapten in close proximity to a chemical functional group or a charged substituent is placed that is expected to transform the corresponding substrate to product. The haptenic charge is then expected to induce a complementary charge at the active site of the antibody. The charged amino acid residues thereby are chosen to contribute to catalysis as general-acid/base or nucleophilic catalysts. Since the haptens designed according to this strategy serve as ‘bait’ for eliciting catalytic functions during the immunization process, which is then ‘switched’ for the substrate, the strategy is called ‘bait-and-switch’.
Using this strategy, Antibody MATT.F-1, raised against a quaternary ammonium hapten 31 whose TSA hapten is 30, the hydrolysis of phosphodiester bonds (which are often found in DNA and RNA and is a reaction of significant importance in living systems) was achieved with high proficiency (Figure 3.37). The Designed Hapten 31 incorporate a general base in an antibody binding site proximal to the 2′ hydroxy of substrate 32 to facilitate nucleophilic attack of this hydroxyl group on the adjacent phosphoryl center. Thus, here the transition state mimicry is sacrificed and replaced by a point charge. MATT.F-1 has a catalytic proficiency [(kcat/Km)/kuncat] of 1.6 × 107 M−1, which is higher than that reported for antibody 2G12, elicited to the transition state analog hapten 30. However, the proficiency of MATT.F-1 is only three times lower than that of the naturally occurring enzyme RNAseA for the same substrate for same hydrolysis.
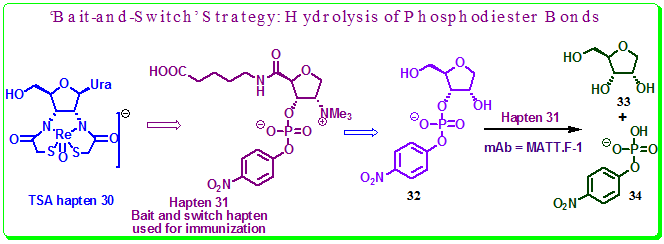
Figure 3.37: Hapten 31 (Bait and Switch) and the hydrolysis of phosphodiester bond catalyzed by mAb MATT.F-1.