3.12.3.3. The Reactive Immunization Strategy
It is clear now that both the TSA and bait-and-switch strategies to generate catalytic antibodies rely for the most part on chemically inert antigens. However, Antigens that mimic the geometric and/or electronic features of a reaction’s transition state can enable the generation of catalytic antibodies. Therefore, a new hapten design strategy––reactive immunization––may provide a chance for eliciting catalytic antibodies to realize the catalytic efficiency of natural enzymes through the use of reactive immunogens.
Thus, to explore this strategy, in 1995 Janda, Lerner and co-workers designed an organophosphorus diester hapten 35, as the primary reactive immunogen to elicit catalytic antibody to perform a chemical reaction in the antibody-combining site during immunization. This hapten can be either hydrolyzed at physiological pH or trapped by a nucleophile at the B-cell level of the immune response affording the monoester 36, an analog of the transition state (Figure 3.38A). Nineteen monoclonal antibodies (mAbs) were isolated, out of eleven were able to catalyze the target acyl-transfer reaction, hydrolysis of phosphonate diester 37 to generate the monoester 38 (Figure 3.38C). Amongst these eleven catalytic antibodies, SPO49H4 demonstrated the best catalytic activity. It effectively catalyze the hydrolysis of the activated ester 39 to yield carboxylic acid 40 with a kcat of 31 min−1 and a rate acceleration (kcat/kuncat) of 6700 at pH 8.0.
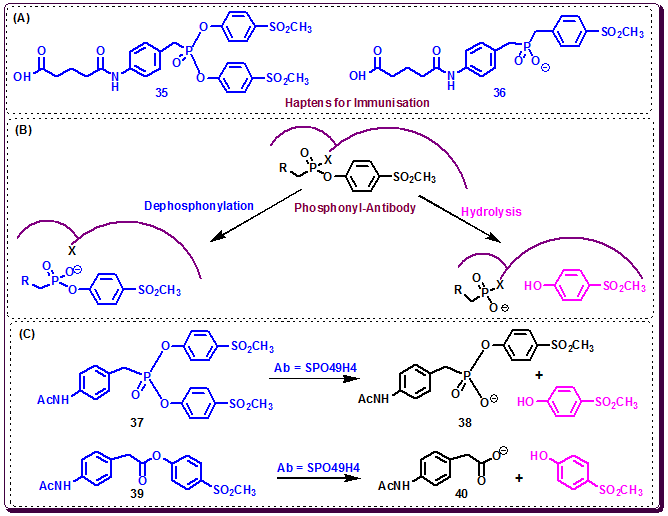
Figure 3.38: (A-B) Haptens and possible chemical reaction that can take place in the antigen combining site when the antibody reacts with the phosphonate diester [X = nucleophile in the antibody]. (C) Reactions catalyzed by catalytic Ab SPO49H4.
The first direct comparison between reactive immunization and transition state analog hapten for catalytic antibody production was demonstrated by comparing esterase antibodies elicited against a TSA, phosphonate monoester 45, with the hapten raised by a reactive immunogen, phosphonate diester 44. Thus, hapten 44 was initially design for the purpose of resolving a racemic mixture of naproxen esters. Antibody 15G2 generated by using hapten 44 (reactive immunization) catalyzed the homochiral production of the anti-inflammatory agent Naproxen, rac-42, from 41. The S-(+)-enantiomer 42a of naproxen is formed 28 more times than the R-(−)-enantiomer 42b (Figure 3.39). This antibody catalyzed the hydrolysis of S-(+)-41a and gave S-(+)-42a with a kcat = 28 min−1, Km = 300 μM at pH 8.0, and kcat/Km = 9.3 × 104 M−1 s−1. On the other hand, antibody 6G6, raised against the transition state analog hapten 45, catalyzed the same reaction with a kcat = 81 min−1, Km = 890 μM at pH 8.0, and kcat/Km = 4.5 × 104 M−1 s−1. The transition state analog approach provided good biocatalysts in terms of turnover numbers and enantiomeric discrimination, but it suffers from varying degrees of product inhibition by phenol 43. Therefore, the antibody generated via Reactive immunization are more efficient and do not suffer from product inhibition.
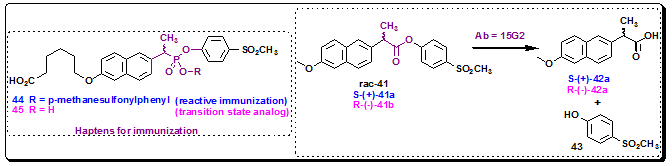
Figure 3.39: Kinetic resolution by catalytic antibodies, Ab 15G2.