The reactive immunization strategy has also been utilized in the aldol reaction. Two aldolase antibodies, 38C2 and 33F12, were elicited by hapten 46, equipped with a reactive β-1,3-diketone functionality. The β-1,3-diketone moiety demonstrated its distinct ability to trap a lysine side chain amine affording an enaminone 48 through Schiff base 47, which directly participates in the mechanism of the aldol reaction in the active site of the antibody (Figure 3.40). The two antibodies, 38C2 and 33F12, obtained by reactive immunization performed extraordinary catalysis of the aldol reaction between acetone and the aldehyde 49, with high catalytic proficiency (Figure 3.40).
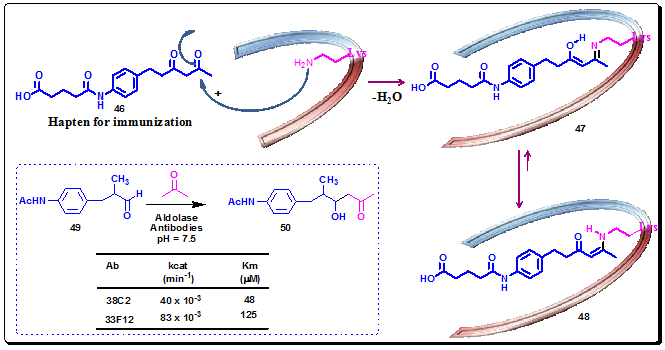
Figure 3.40: Mechanism of trapping the essential e-amino group of a lysine residue (LysH93) in the antibody’s binding pocket by using the 1,3-diketone hapten 46 and Aldol reaction catalyzed by mAb 38C2 and 33F12.
Zong et al. employed reactive immunization strategy in combination with transition state theory to find more efficient aldolase antibodies. Thus, on the basis of hapten 46, a hybrid, hapten 51, was designed, employing a sulfone functionality to establish the tetrahedral motif that present in the transition state, and a β-diketone for trapping a lysine side chain at the active site (Figure 3.41). Two aldolase antibodies, 93F3 and 84G3 were isolated. In the aldol reaction of 54 with 3-pentanone, antibody 93F3 provided syn-aldol 55 with 90% de and 99% ee, while antibody 38C2, elicited to hapten 46 afforded only 62% de and 59% ee. Antibodies 93F3 and 84G3 showed a 103-fold increase in proficiency over the antibody 38C2 for the kinetic resolution of (±)-56(Figure 3.42) [Zhong, G. F.; Lerner, R. A.; Barbas, C. F. Angew. Chem., Int. Ed. 1999, 38, 3738].
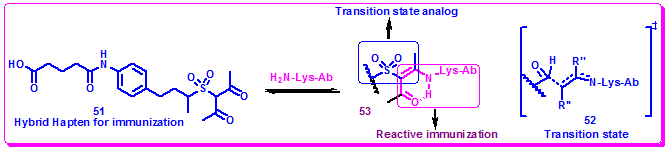
Figure 3.41: The hybrid hapten 51 designed from the presume transition state.
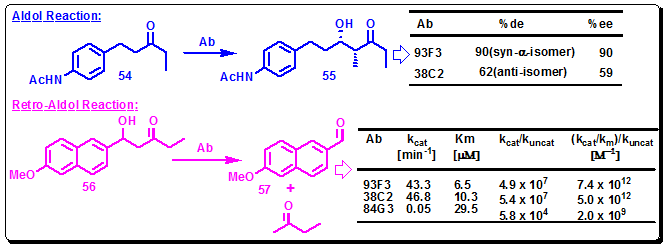
Figure 3.42: Comparison between 93F3, 84G3, and 38C2 for aldol and retro-aldol reactions.
Mechanism-based inhibitors covalently react with the active site in target proteins and inhibit their activities. Therefore, such inhibitors provide a wealth of information to guide the design of haptens for immunization for generating catalytic antibodies. Penam sulfones are potent mechanism-based inhibitors of β-lactamase. They by forming an acyl-enzyme intermediate inactivate the enzyme. Therefore, this efficiency of sulphones inspired the design of the sulfone hapten 58 targeting the hydrolysis of the lactam functionality built in the substrate 59. Immuno conjugate 58-KLH was immunized, and a scFv library was constructed using the spleen cells of immunized mice. Screening of the library yielded two scFv antibodies, FT6 and FT12. These antibodies catalyzed hydrolysis of 59 with rate accelerations (kcat/kuncat) of 5200 and 320, respectively (Figure 3.43).
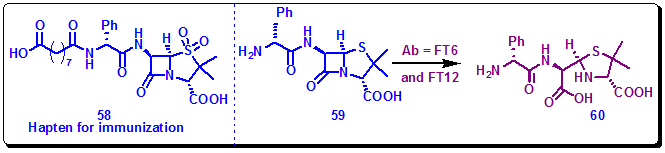
Figure 3.43: Reactive hapten 58 and the reaction catalyzed by mAb FT6 and FT12.