3.10.3. Enzymes Are a Distinct Target Class
Enzymes are biocatalysts. Among all other catalytic mechanisms shown by enzymes as was discussed earlier under the heading of “mechanism of enzyme catalysis” in this module, they ability of formation and break down of specific covalent chemical bonds makes enzyme a unique drug target and different from, Cell surface receptors, ion channels, transporters, nuclear hormone receptors, and nucleic acid/ribosome target. In drug discovery, the focus is always on the binding event of the drug with the enzyme target. Enzyme catalysis progresses through binding events, conformational changes, one or more transition states, or reaction intermediates, and product release, and all of these steps occur with defined rate constants. The rate constants define a thermodynamic profile that can be used for drug design, and this differentiates enzymes from all other target classes.
Drugs regulate the rate of chemical reactions of an enzyme. Many of most important and powerful modern drugs act on enzymes in the plasma or inside cells. Their action is usually via enzyme blockade and examples include the angiotensin I-converting enzyme (ACE) inhibitors. Almost all biological reactions are carried out under catalytic influence of enzymes which are the major drug target. Drug may increase (stimulate) or decrease the activity of enzymes. However, enzyme stimulation is less common by drug. Generally, drugs inhibit the enzyme which is the common mode of action of drug and is the best approach for a drug design. We have passed through several kinds of enzyme inhibition and accordingly drugs are designed. There are mainly the following types of drugs which target enzyme.
- Enzyme Substrate Analogues/Inhibitor as Enzyme-Targeted Drugs
- Transition-State Inhibitors as Enzyme-Targeted Drugs
- Irreversible Inhibitors as Enzyme-Targeted Drugs: The following 25 enzymes are irreversibly inhibited by drugs.
(1) Serine Type d-Ala-d-Ala Carboxypeptidase; (2) β-Lactamase; (3) Acetylcholinesterase; (4) UDP-N-acetylglucosamine 1-Carboxyvinyltransferase (Fosfomycin); (5) Prostaglandin-Endoperoxide Synthase (Aspirin); (6) Unspecific Monooxygenase; (7) Amine Oxidase (Flavin-Containing); (8) Thymidylate Synthase; (9) Ornithine Decarboxylase; (10) Alanine Racemase; (11) H+/K+ ATPase; (12) Triacylglycerol Lipase; (13) Ribonucleoside-Diphosphate Reductase; (14) Iodide Peroxidase; (15) Thyroxine 5‘-Deiodinase; (16) Aldehyde Dehydrogenase; (17) Thrombin; (18) Factor Xa; (19) 4-Hydroxyphenylpyruvate Dioxygenase; (20) Vitamin K Epoxide Reductase; (21) Ile tRNA Synthetase; (22) DNA-Directed DNA Polymerase; (23) 3-Oxo-5-α-steroid 4-Dehydrogenase; (24) Enoyl-Acyl Carrier Protein Reductase; (25) Xanthine Oxidase.
- Reaction Intermediate Traps as Enzyme-Targeted Drugs
- Boronic Acid-Based Inhibitors as Enzyme-Targeted Drugs
- Noncompetitive Inhibitors as Enzyme-Targeted Drugs
- Activators as Enzyme-Targeted Drugs
3.10.3.1. Enzyme Substrate Analogues/Inhibitor as Enzyme-Targeted Drugs
The majority of marketed enzyme-targeted drugs are related to enzyme substrate structure. These drugs, either undergo catalysis in the active site of an enzyme, chemically react with an enzyme cofactor, or contain a structural motif related to the substrate. Antibiotics, including penicillins, cephalosporins, and carbenapems, that target serine type d-Ala-d-Ala carboxypeptidase, and they all bear some structural similarity to the terminal d-Ala-d-Ala of bacterial peptidoglycan. In addition, they all undergo catalysis on the enzyme and acylate the active site serine. Similarly, inhibitors of β-lactamase, are used to overcome β-lactamase resistance by acylating the β-lactamase active site serine.
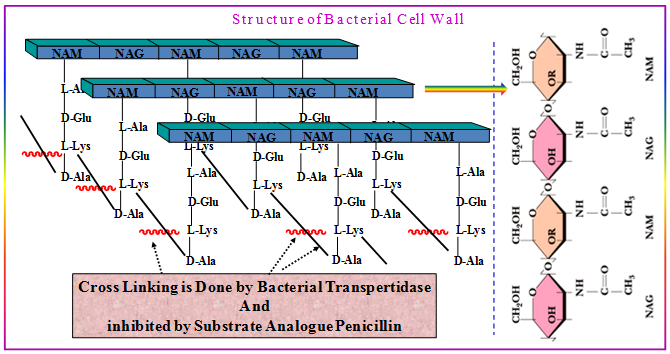
Figure 3.6: Structure of Bacterial cell wall.