They have synthesized novel enantiopure (i)-(β-lactam)-(Gly)-(i+3) peptide models starting with β-lactam scaffolds, defined by the presence of a central α-alkyl-α-amino-β-lactam ring placed as the (i+1) residue. In their conceptual design, while the β-lactam methylene group mimics simultaneously the CαH(i+1) and HN(i+2) protons of the native peptide, R1 and R2 groups can be designed specifically for recognition with the receptor (Figure 2.55). In this approach, it was also assumed that the presence of the α,α-disubstitution pattern would enhance resistance to chemical and enzymatic hydrolysis by proteases and that the resulting β-lactam-peptides would be attractive targets for pharmaceutical drug discovery.
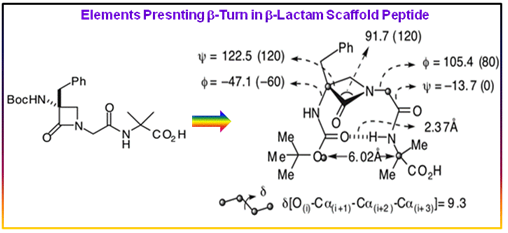
Figure 2.56: β-lactam based peptidomimetic indicating a type-II' β-turn conformation.
The structural properties of these b-lactam pseudopeptides were studied by x-ray crystallography, molecular dynamics simulation, and NOESY-restrained NMR simulated annealing techniques, showing a strong tendency to form stable type II or type II' β-turns either in the solid state or in highly coordinating DMSO solutions. Crystal structure of the peptidomimetic (Figure 2.56) indicated a type-II' β-turn conformation.
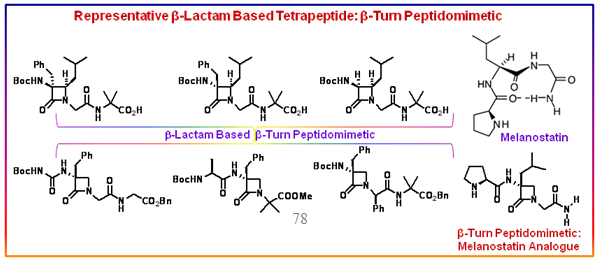
Figure 2.57: Tetrapeptide models as a function of the structural and stereochemical elements of the β-lactam core for β-turn induction.
Tetrapeptide models containing syn- or anti-α, β-dialkyl-α-amino-β-lactam rings were also synthesized and their conformations analyzed, revealing that α-alkyl substitution is essential for β-turn stabilization. A β-lactam analog of melanostatin (H-Pro-Leu-Gly-NH2) was prepared. The compound (Figure 2.57) was characterized as a type-II β-turn in DMSO-d6 solution, and was tested by competitive binding assay as a dopaminergic D2 modulator in rat neuron cultured cells, displaying moderate agonist activity in the micromolar concentration range.
Biological Evaluation of Melanostatin (PLG) β-Lactam Analogue: Several studies have shown that PLG render the Gi-protein coupled dopamine D2-like receptors more responsive to agonists by maintaining the high-affinity binding state of the receptor. Though the full mechanism is not clear, but it is known that the mechanism of action of PLG may likely involve intraneuronal enzymes. So, direct competitive binding assays on integral neurons can provide information more directly assignable to real living systems.
To test the relative activity of β-lactam PLG analogue versus natural PLG as a dopamine D2 receptor modulator, Palamo et al. have conducted a radio ligand competitive binding assays on cultured integer neuron cells from rat cerebral cortex. Their competitive binding assay study indicated that β-lactam PLG analogue (Figure 2.57) slightly reduces the dissociation constant of dopamine D2 receptors for N-propylnorapomorphine (NPA) with respect to PLG at a micromolar concentration range. The full retention of bioactivity on living neurons observed for β-lactam PLG analogue with respect to natural PLG suggests that the structural variation arising from the replacement of CαH(i+1) and HN(i+2) protons by a β-lactam methylene group does not hinder the maintenance of a similar recognition pattern by D2 receptors.