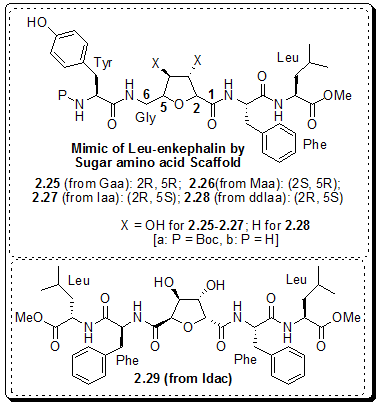
Figure 2.49: Leu-enkephalin mimetic by Chakraborty et al.
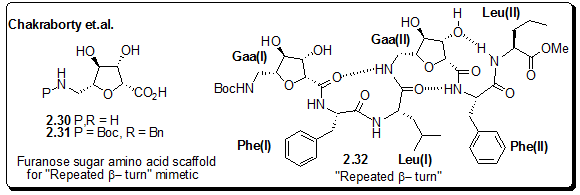
Figure 2.50: Repeated b-turn mimetic by Chakraborty et al.
A new reverse turn, replacing one of the native type II' β-turns in the cyclic peptide antibiotic gramicidin S, induced by a furanoid sugar amino acid was recently reported by Grotenbreg and coworkers. NMR and X-ray crystallographic analysis showed that C3-hydroxyl function plays a pivotal role by acting as an H-bond acceptor, consequently flipping the amide bond between residues i and i + 1.
Recently, Ferrocene derivatives were proposed as an organometallic scaffold for β-turns. Herrick et.al, in 1996, reported the compounds of general structures Fe(C5H4-CO-Xaa-OMe)2 (with Xaa = amino acid) to have an ordered structure in organic solvents and this structure is stabilized by two symmetrically equivalent hydrogen bonds between the amide NH and the methyl ester carbonyl moiety of another strand, 2.34 (Figure 2.51). Hirao and coworkers prepared several substituted dipeptides of similar ordered structure.
Cobaltocene dicarboxylic acids have also been used as a scaffold for β-turn mimetic. Recently, von Staveren et.al. have investigated the influence of a positive charge on the structure and stability of peptide turn structures, which are stabilized by H bonds. They have concluded that turn structure in 2.35 is g-turn like and in 2.36 is β-turn-like (Figure 2.51). Because of parallel orientation induced by the diacid scaffold, they have designated the turn structures as pseudo-gp and pseudo-βp turn.
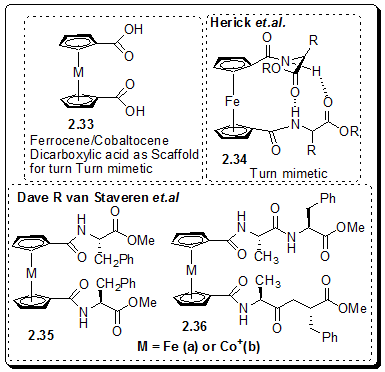
Figure 2.51: Peptide β-turn mimetic based on ferrocene/cobaltocene dicarboxylic acid scaffold
2.6.4. Z-Enediyne π-chromophores As Possible Scaffold for Peptidomimetics Design
During the past decade, the construction and investigation of expanded acetylenic p-chromophores has become a central area of chemical research. It has been fueled by the availability of new synthetic methods, in particular Pd(0)-catalyzed cross-coupling reactions, the discovery of the antitumor activity of a series of natural compounds possessing reactive Z-enediyne π-chromophores, and the need for new nanoscale molecules and polymeric materials that exhibit unusual electronic and optical functions and properties. The enediyne antitumor antibiotics are appreciated for their novel molecular architecture, their remarkable biological activity and their fascinating mode of action and many have spawned considerable interest as anticancer agents in the pharmaceutical industry. Of equal importance to these astonishing properties, the enediynes also offer a distinct opportunity to study the unparalleled biosyntheses of their unique molecular scaffolds and what promises to be unprecedented modes of self-resistance to highly reactive natural products. Elucidation of these aspects should unveil novel mechanistic enzymology, and may provide access to the rational biosynthetic modification of enediyne structure for new drug leads, the construction of enediyne overproducing strains and eventually lead to an enediyne combinatorial biosynthesis program.