2.7 β-Lactam Based Peptidomimetics
Templated β-turn peptidomimetic is a complex task in respect of the synthesis of molecular templates with precisely oriented predetermined groups. A great deal of effort has been dedicated to the development of such peptidomimetics. A rational design of an efficient mimetic necessitates the modification of the native bioactive peptide to include two major elements-(a) a specific β-turn constraining element such as an intra- or inter residual bridge, is needed to force the peptide backbone to adopt the desired conformation as exactly as possible and (b) at least one recognition group must be placed in a stereocontrolled fashion at the desired position for interaction with the receptor or enzyme active site.
Though it is simple to design, practical access to peptidomimetic libraries bearing a broad range of recognition groups, while retaining the β-turn framework, often is a tedious and synthetically challenging job starting from the scaffold chosen. Also, it is difficult or impossible to elucidate structural elements exerting specific constraints or recognition functions for most known β-turn surrogate molecules. As a consequence, chemical alteration of such peptidomimetics, when synthetically feasible, leads to unpredictable results in terms of β-turn motif stability and/or proper spatial orientation of the recognition groups.
An important example of this phenomenon (Figure 2.54) is the original approach of Freidinger to β-turn mimetics. The Freidinger’s original approach to β-turn mimetic, involved the bridging of the betagenic -(i+1)-(i+2)- central residues through the formation of five-, six-, seven-, and eight-membered ring lactams. Accordingly, any structural modification of the R1 bridge in pseudopeptide (Figure 2.54), should force a global change of conformation and recognition properties that, in practice, involves a re-design of each derivative prepared, instead of a single structural parameter variation.
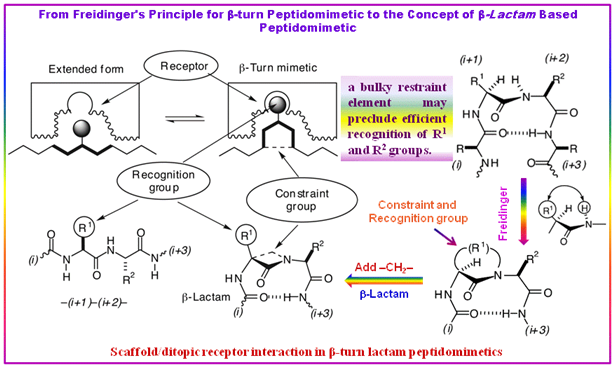
Figure 2.54: The designing concept of β-lactam based peptidomimetics.
The Freidinger’s idea of β-turn peptidomimetics based on the principle of the incorporation of groups, as small as possible, in the original peptide to generate well-defined turns without affecting receptor recognition was explored by Claudio Palomo et al. in designing the β-lactam-based peptidomimetics as a novel family of β-turn motif nucleators. This concept was illustrated by their designed β-lactam peptides and β-lactam peptidomimetics of melanostatin (PLG).
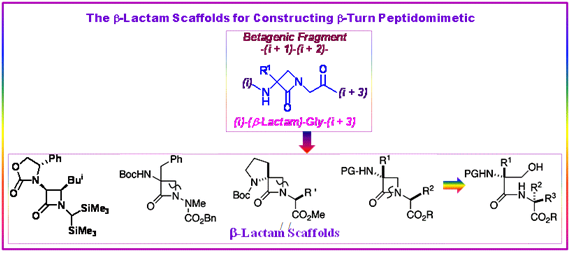
Figure 2.55: β-Lactam scaffolds for the peptidomimetics; Palomo et.al.