2.6.5. Enediynyl Amino Acid AS β- Sheet Nucleator
As already mentioned, Enediynes have drawn unprecedented interest amongst the scientific community because of their cytotoxic activity and possible use as anticancer drug. All studies so far have been concentrated on their synthesis and evaluation of chemical as well as biological activity. The special structural feature of Z-enediynes is the type of reverse-turn associated with the two acetylenic arms. One can consider making enediynyl amino acid containing peptides, which may be forced to adopt typical conformational motifs.
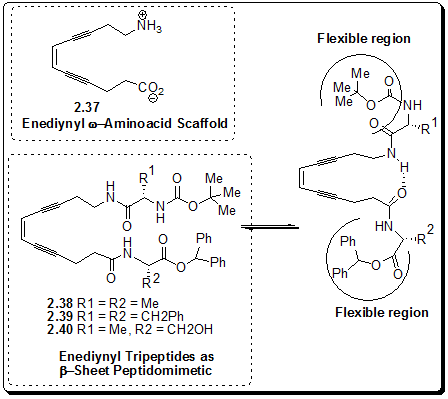
Figure 2.52: The solution conformation of the enediynyl peptides
This structural motif, for the first time in enediyne chemistry, was used, in our laboratory, as a possible scafolld for peptide secondary structure mimetic. Specially, we were interested in designing and synthesis of the b-sheet mimetic by incorporating the w-enediynyl amino acid into a peptide chain. We thought that this enediynyl motif can act as nucleator and thus may induce in adopting the b-sheet which constitutes a well-studied subset of the reverse turn and is a common feature in biologically active peptides and globular proteins. The sheet capping turns are widely believed to act as a molecular recognition site for many biological processes.
Bag and Basak et al. have incorporated the enediynyl amino acid 2.37 into peptides 2.38-2.40 (Figure 2.52) and then found out the conformational preferences by NMR and CD-measurements.
Circular Dichroism (CD) spectra of the fully protected peptides and the generally higher (Δd/δT) values for the chemical shifts of β and γ N-H’s reveales that the peptides adapt a significant proportion of β-sheet like conformation. However, the results also indicated the presence of other conformations as well, specially the a-NH as being intramolecularly H-bonded. The variable temperature NMR experiments indicate that the conformation resembling β-sheet capping type motif is more predominant. The situation is represented in Figure 2.52.
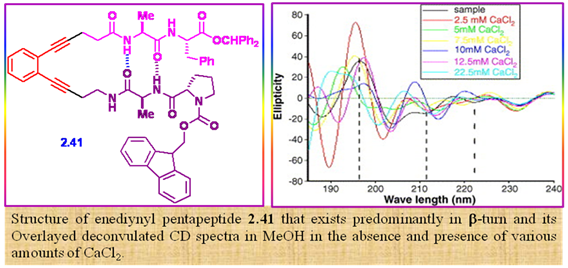
Figure 2.53: The enediynyl pentapeptide and its CD spectra.
A novel enediynyl pentapeptide in the protected form 2.41 was synthesized and characterized. It exists predominantly in β-turn structural motif as revealed by variable temperature. NMR and CD spectroscopy. In the presence of transition metal ions and gold nanoparticles, the fluorescence intensity of the peptide got enhanced with remarkable quantum yield with the Z-enediynyl ω-amino acid acting as a fluorophoric reporter. The interesting photophysical behaviors with alkali and alkaline earth metal ions are also reported.
The secondary structure of peptide 2.41 was estimated by recording its CD spectrum in methanol, which showed a strong maximum at ∼198 nm followed by several broad minima at ∼205, 212, and 222 nm, indicating that the peptide predominantly adopts a β-turn like structure (Figure 2.53) at least in the solvent used for the study. The peptide secondary structure estimation using CD estima program shows a 60% turn like structure, the existence of which implies the possible presence of intramolecular H-bond between the peptide strands on the two arms of the enediyne framework. This could be assessed by determining the variation of chemical shifts of the various NHs with temperature in DMSO-d6 in which all the four NHs exhibited different chemical shifts. Interestingly, the turn like structure is more or less maintained in the presence of Ca2+ ions.
Of the four amide NH’s, one alanine NH and the NH belonging to the enediynyl amino acid exhibited Δδ/ΔT values that are within the Kessler limit of −3 ppb/K, indicating strong intramolecular H-bonding and supported the predominant turn like structure of the peptide. The appearance of a crosspeak for the hydrogens attached to C-2 and C-11 in NOESY spectrum also provided further evidence for the turn like conformation of the two peptide arms of the enediyne backbone. The H-bonded conformation was also supported by the semi-empirical AM1 geometry optimization.