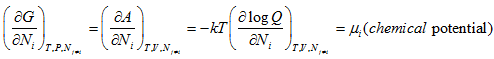By combining the first and second law of thermodynamics we get:
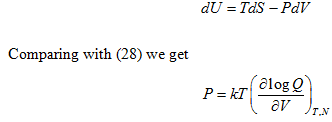
Also
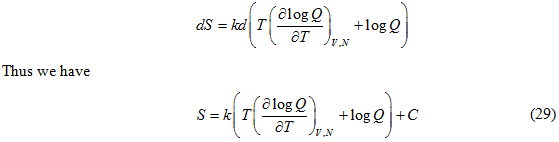
The constant C turns out to be zero since at absolute zero i.e at T=0 K we have S=0 (third law of thermodynamics). Thus the entropy is given by:
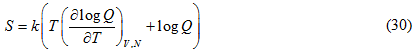
In a similar manner we can obtain the expression for Helmholtz free energy i.e
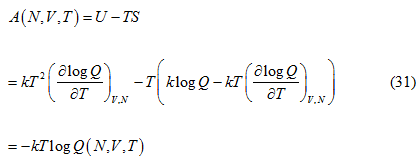
This also follows the equivalence among Gibb’s free energy(G), Helmholtz free energy(A) and the chemical potential and is given as: