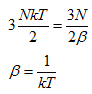Partition Functions of Ideal Monoatomic Gas
Till now the relation between β and Temperature is unknown. Let us consider a collection of non-interacting particles in a closed box so that it can be treated as an ideal monoatomic gas.The monoatomic gas has been taken since the relation between β and Temperature obtained will remain valid for all type of systems. The derivation of the single particle of the monoatomic gas may be found in many textbooks and hence it is now shown.

Where m is the mass of one particle and h is the Planck’s constant. Thus now writing out the total partition function we get
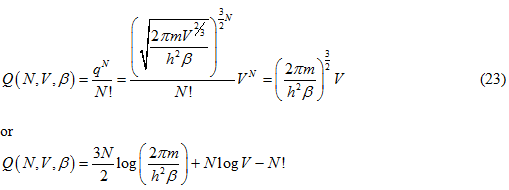
Thus the internal energy of N particle system is given by:

If we look closely equation (a) and apply the ergodic hypothesis, the internal energy must be equal to that for an ideal gas. The internal energy of an ideal monatomic gas by definition is equal to ![]() .Thus comparing this relation with equation (24) we get:
.Thus comparing this relation with equation (24) we get: