Mechanisms of few acid-base catalyzed processes are discussed below.
Ester hydrolysis: The hydrolysis of esters is catalyzed by both acid and base. H2O act as the proton donor. The mechanism for acid and base catalyzed hydrolysis can be represented as:
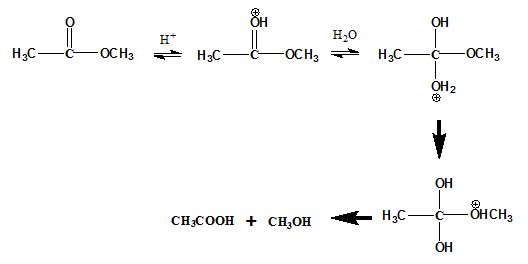
Acid catalyzed hydrolysis:
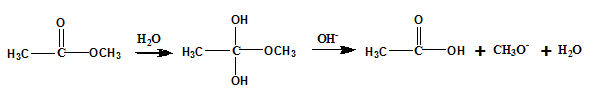
Base catalyzed hydrolysis :
Dehydration: Acid catalyzed dehydration mechanism is illustrated by dehydration of ethyl alcohol to ethene. Alcohol is initially protonated. Then water molecules leaves forming a carbocation . Then ß-elimination occurs producing the alkene .
![]()
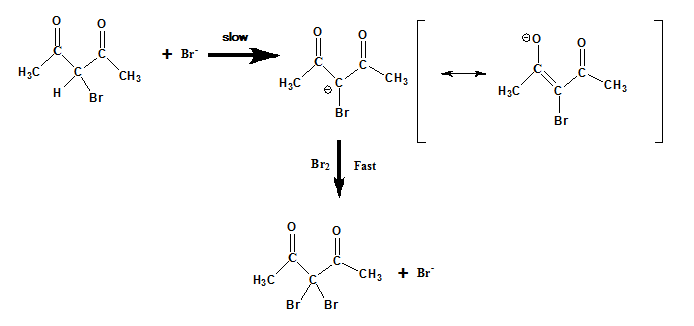
Halogenation : For base catalyzed halogenation reactions, the probable reaction mechanism is illustrated below. The ionization of the ketonic substrate in the presence of basic catalyst is the rate determining step.
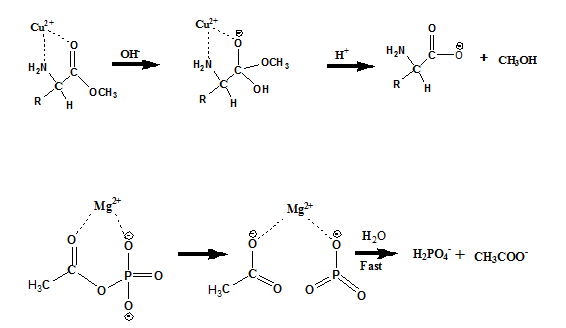
Catalysis by Metal ions
The metal ions can coordinate simultaneously to electron donating atoms, such as N and/or oxygen, present in the reactant. The probable mechanism of Cu2+ and Mg2+. ion catalyzed hydrolysis are shown below.