Homogeneous catalysis mechanism is often represented by catalytic cycles. In catalytic cycles usually catalysts are shown as member of cycle and all reactants and products are placed outside the cycle and connected to it by arrows.
For example the cycle of a catalytic reaction A+B →P, having intermediates K and L can be represented as
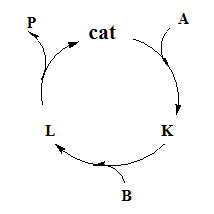
The stable metal complex added to the reaction at the beginning is called catalysts precursors or precatalysts. Turnover frequency in terms of catalytic cycle can be defined as number of times the cycles is completed in unit time.
The intermediates species can be studied using various spectroscopic studies such as FTIR, NMR, UV-Vis etc.
Acid -base catalysis
Mechanism
In acid-base catalysis both acid and bases may act as catalysts in solutions. The H+ is used for protonating the intermediates and a base or solvent is used for removing the proton at later stage. The Rate of acid –base reactions depends on pH since the rate is a function of both H+ and OH− concentrations. The proton transfer mechanism in general can be represented as
![]()
Kinetics
In acid catalysis, the proton transfer step is slower and is the rate determining step. Subsequently, the protonated substrate rapidly reacts to give the product(s) as shown below :
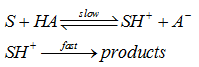
Reaction rate is dependent on all acids/bases present in solution. Rate constant for reaction is function of concentrations of H+, OH−, HA and A− . First order rate constant can be calculated from
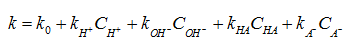
Where k0 = rate constant for uncatalyzed reaction (small relative to other terms)
Activity of acid-base catalyst
Activity of an acid depends on its acid strength and molecular structure. Acids of higher strength are more catalytically active. Strength is defined in terms of equilibrium constant (KHA) for dissociation. ![]()
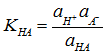 where ai is the respective activity
where ai is the respective activity
For general acid catalyst, rate constant depends on its acid strength as :
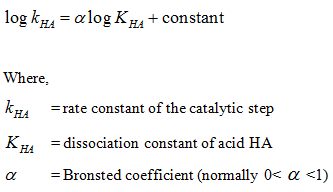
α indicates the sensitivity of catalytic step for changes in acid strength of HA. Similarly Brønsted relation for general base catalysis is
![]()
The coefficient β has the same meaning as α for general acid catalysis.