Mechanism and reaction rate
Activity
The homogeneous catalyst precursors are added in the reaction system in different forms and are transformed into the active form insitu. During one catalytic cycle, the catalyst may pass through several intermediate forms and finally produce the products. After end of each catalytic cycle, the catalyst itself should be regenerated without any change.
A catalyst should be able to pass through the catalytic cycle multiple times. Higher the number of times the catalyst passes through this cycle, higher is the activity of the catalyst. The number of times that a catalyst can go through this cycle converting substrate molecule to product molecules is defined as the turnover number. In other words, the turnover number, TON, is the total number of substrate molecules that a catalyst can convert into product molecules. In homogeneous systems, the turnover frequency is defined as the number of molecules of substrate converted per second which is the turnover number in a certain period of time.
Selectivity
Following type of selectivities are defined :
- Chemoselectivity
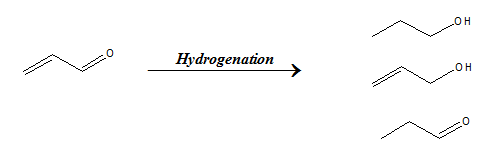
When two chemically different functionalities are present in same molecule, then the selective reaction of one functional group in presence of the other is known as chemoselectivity. As shown below, the alkene and aldehyde groups present in the same molecule can undergo hydrogenation resulting in products I, II or III as a result of hydrogenation of both groups, only aldehyde or only alkene respectively. Selectively hydrogenating any one of the two functional groups and maximizing the corresponding product is known as chemoselectivity.
- Regioselectivity
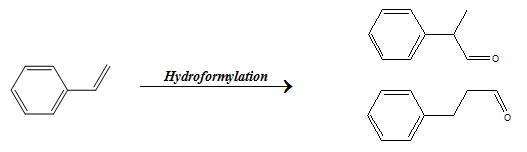
When the functional group can attach to multiple sites, then the selectivity determining the site of attachment is known as regioselectivity. As in the example shown for the hydroformylation reaction of styrene in which the formyl group can attach to either terminal carbon atom or secondary internal carbon atom resulting in linear or branched product respectively.
- Diastereoselectivity
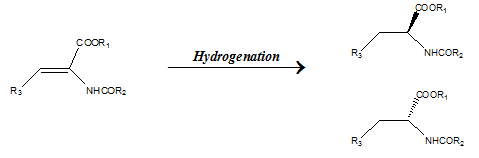
The isomeric molecules having similar molecular formula but differing in the three-dimensional orientation of atoms are known as stereoisomers. Diastereomers are two or more stereoisomers of a compound that are not mirror images of each other. When stereoisomers are mirror images of each other that are non-superimposable, they are called enantiomers.
For a substrate containing a stereogenic centre the catalysts can direct the addition of atoms to give two diastereomers. The selectivity for either diastereomer is called diastereoselectivity. For example the addition of dihydrogen as shown below gives two diastereomers.
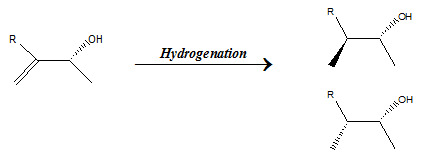
- Enantioselectivity When substrate is achiral, that is the molecule is superimposable on its mirror image, catalyst may give rise to the formation of specific product enatiomer as shown in the example below. This is known as enantioselectivity.