Water gas shift reaction
Water gas shift reactions are carried out at conditions of high and low temperature. For the water gas shift reaction, the equilibrium favors the reaction at lower temperature. However, copper based catalyst, active at low temperature, is highly sensitive to residual sulfur or chloride compounds which have passed through the pre-purification steps. Hence, initially the reaction is carried out over catalyst composed of Fe2O3 and Cr2O3 which is relatively inexpensive, resistant to sintering and tolerant to sulfur and chloride poisoning. However, at low temperature Fe3O4 is not sufficiently active hence, the reaction has to be carried out at higher temperature. The high temperature catalyst adsorbs the residual sulfur or chloride compounds thereby, protecting the low temperature copper based shift catalyst from exposure to these compounds. The high temperature process is followed by shift reaction at thermodynamically favorable lower temperature of 200°C over copper based catalyst.
High temperature water gas shift reaction
Gas from the secondary reformer contain 10-13 % CO and is further processed to increase the H2 concentration. The high temperature water gas shift reaction lowers the CO concentration to about 2-3 % in a fixed bed reactor.
![]()
Process is operated at 350-5000C and 20 -30 atm. Since equilibrium favors the WGS at lower temperature, the feed gas is cooled to 350-4000C before entering the reactor.
Catalysts:
A typical catalyst is composed of 90 % Fe3O4 and 10 % Fe2O3. Fe3O4 acts as the active metal while Fe2O3 acts as a stabilizer to minimize sintering of the active iron oxides. The catalysts are prepared by precipitation.
Kinetics
The rate expression from Langmuir Hinshelwood model proposed is given as [4] :
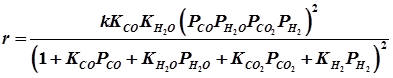
k is the rate constant and Ki's the respective adsorption constants. The ‘i' represents the chemical species.
Activation energy for the water gas shift reaction on iron oxide/chromia is in the range of 122 kJ/mol. Reaction over small pellets (5 x 4 mm) operates with an effectiveness factor of unity below 3700C and 31 atm.
Deactivation
The catalyst is deactivated by adsorption of residual S or Cl containing compounds that escape the initial purification steps.
Low temperature water gas shift
The CO concentration of the existing gas from high temperature WGS is 2-3 % and further reduced to below 0.2 % by low temperature water gas shift reaction. The WGS reaction as favored at low temperature, operating temperature is maintained at 2000C, the minimum temperature at which steam is not condensed and operating pressure at 10-30 atm.
Catalysts
A typical catalyst is composed of CuO/ ZnO/Fe2O3 and prepared by co-precipitation method. The catalyst is highly selective for shift reaction with low activity for methanation reaction. CuO is the catalytically active metal and ZnO minimize sintering of Cu. Cu is highly sensitive to poisoning by S or Cl compounds even at very low level of 1 ppm. ZnO & Fe2O3 also scavenge the S or Cl compounds protecting Cu which is highly sensitive to poisoning by these compounds. Catalyst is reduced very carefully by H2 and the bed temperature is never allowed to rise above 2300C to avoid sintering of Cu. The high temperature shift catalyst also protects the low temperature catalyst from these poisons.
Kinetics
Kinetic m odel is based on surface redox mechanism involving dissociation of water to OH radicals which further dissociate to atomic oxygen. The rate expression as proposed by Ovensen et al.[2] is
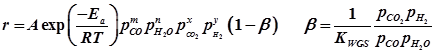
A= pre-exponential factor; Ea= activation energies; m, n, x, y = respective reaction orders; β defined as approach to equilibrium and KWGS is equilibrium constant for WGS reaction.
Final CO/CO2 removal by methanation
After low temperature WGS, the CO content in hydrogen stream is 0.2-0.5 % and must be reduced to 5 ppm level for using the H2 for ammonia synthesis. The CO is further reacted with hydrogen and converted to methane. The catalyst is typically Ni/Al2O3. MgO is sometimes used as a promoter to minimize sintering of Ni. The process is carried out at 300 -3500C and at 30 atm pressure. Care must be taken to avoid formation of poisonous Ni(CO)6 which occurs below 2000C at partial pressure of CO greater than ~ 0.2 atm.
Book reference:
• C. H. Bartholomew and R. J. Farrauto, Fundamentals of Industrial catalytic Processes, Wiley, VCH, 2006
Publication reference:
- 1. M.H Halabi M.H.J.M de Croon, J. van der Schaaf, P.D. Cobden, J.C. Schoulen, Applied catalysis A Gen. 389 (2010) 80-91
2. J. Agrell, H. Birgersson, M. Boutonnet, Journal of Power Sources 106 (2002) 249–257
3. M. Zeppieri, P.L. Villa, N. Verdone, M. Scarsella, P. De Filippis, Applied Catalysis A: General 387 (2010) 147–154
4. J.R . Rostrup –Nielsen, Catalysis Science and technology eds. J.R. Anderson and M. Boudart, Springer –Verlag, Newyork,1984
5. T. Numaguchi and K.Kikuchi, Chemical Engineering Science 43(1988) 2295-2301