Process
A schematic diagram depicting the typical reaction conditions in primary steam reforming is shown in Fig. 3. For primary steam reforming, the outlet temperature is limited to about 8000C to prevent damage to the reactor. Though the reaction is thermodynamically favored at low pressure, a higher pressure is used to increase the reaction rate and throughput, thereby minimizing the reactor volume.
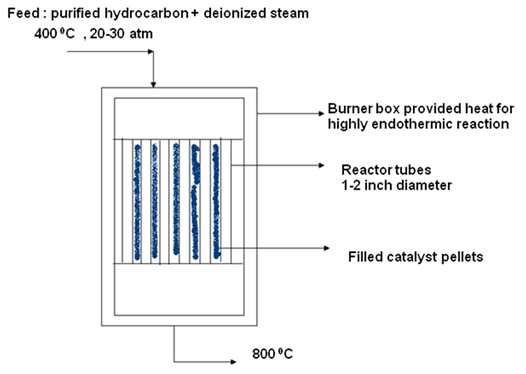
Fig. 3. Schematic diagram showing steam reforming reaction conditions
Operating condition for methane and naphtha steam reforming are very different. For methane steam reforming the reformer exit temperature is typically 800 -9000C and H2O : CH4 ratio is 1.8 -3. In naphtha steam reforming exit temperature is lower 600-8000C and H2O : C ratio is higher 2.5-4.5. In both cases pressure is maintained at 20-30 atm.
Catalysts
Bifunctional catalysts are needed for steam reforming reaction. Two catalytic functions are :
– Metal site for dissociative adsorption of hydrocarbon
– Oxide site for diss ociative adsorption of water
Oxide sites are generally located on the surface of the supports. Support also stabilizes the well dispersed metallic phase. Because of the severe high temperature reaction conditions, catalyst must have high thermal stability. The typical decreasing activity order for metals is Rh, Ru > Ni > Pd, Pt >Ni-Cu >Co. This order varies with hydrocarbon. Pt and Pd are less active than Ni for steam reforming of naphtha, however are more active than Ni for steam reforming of toluene. Noble metals are more resistant to carbon deposition and more tolerant to sulfur poisoning than Ni. However, b ecause of greater availability and lower cost, Ni is the preferred metal catalyst for commercial applications. Thermally stable supports of low acidity are needed to prevent deactivation by sintering and carbon depositions.
The order of decreasing acidity of supports:
![]()
Thus CaO, MgO are most desirable supports to prevent carbon depositions. These are also among most thermally stable supports. Promoters such as potassium alumino - silicate lowers the acidity of supports & catalyze carbon gasification with steam.
Typical catalysts for steam reforming consist of NiO/Ca Al2O4 or NiO/Al2O3. Steam reforming catalysts are chemically and mechanically rugged to maintain physical and chemical integrity in harsh high temperature and high steam environment. Surface area of carrier is low, typically 5-50 m2/g but adequate for dispersing NiO and maintaining thermal stability. Support is impregnated with nickel nitrate solution a number of times to achieve proper loading. Active catalyst is metallic Ni. Therefore NiO after calcination is reduced by H2 prior to reaction. Catalysts are formed into relatively large raschig rings, grooved pellets, wheels etc for low pressure drop and minimum pore diffusion limitations.
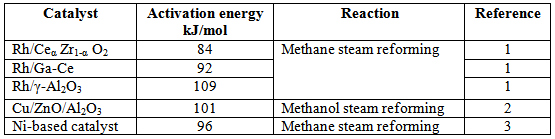
Table 1. Activation energies reported for steam reforming process over various catalysts