Purifications of hydrocarbons
Sulfur and chloride compounds in the feed act as catalyst poisons. Therefore, before the reforming process, feed is treated to remove these compounds. Sulfur is removed by desulphurization followed by scrubbing by ZnO adsorbent. By this process, sulfur is reduced to less than 0.01 ppm
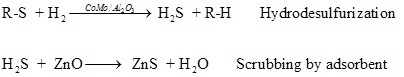
Chloride is removed by scrubbing using alkali treated alumina to 5 ppb level.
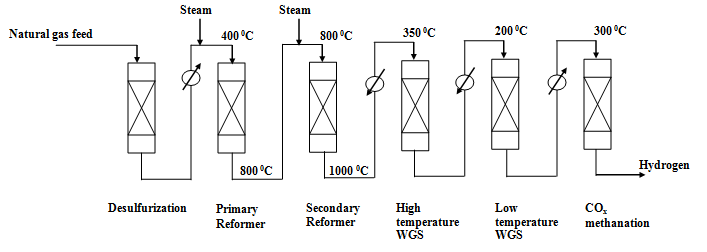
Fig. 2. Schematic flow diagram for hydrogen production from steam reforming of natural gas
Primary steam reforming
For steam reforming of natural gas which mostly consist of methane, the overall reaction is given as:
![]()
This reaction is always accompanied with methanation and water gas shift reaction.
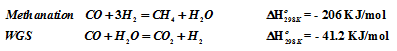
The overall process is endothermic and heat is supplied to the reformer. Relative amount of H2 and CO can be adjusted by process conditions. Since the overall steam reforming reaction is highly endothermic and the product volume is greater than that of the reactants, equilibrium favors the forward reaction at high temperature and low pressure. High steam to methane ratio and high temperature favors conversion of CH4. As temperature is lowered, the slightly exothermic WGS reaction becomes increasingly more favorable. Higher steam to methane ratios at lower temperature result in lower CO concentration. The H2 to CO ratio thus depends on operating temperature for steam reforming and water gas shift reactions.