Kinetics and mechanism
Mechanistically steam reforming involves decomposition of hydrocarbons on metal surface to hydrocarbon fragments. Water undergoes dissociative adsorption to adsorb oxygen atoms and molecular hydrogen. Adsorbed oxygen atom combine with adsorbed hydrocarbon to form CO. The kinetic model proposed by Rostrup and Nielsen [4] is as follows
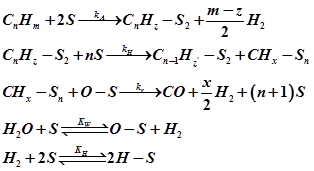
The corresponding rate was derived as
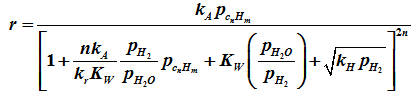
Industrial steam reforming reaction is strongly influenced by pore diffusion resistance and therefore apparent activation energies are lower compared to surface reaction controlled system. Activation energy for nickel based catalyst for steam reforming reaction 106 kJ/mol and that for water gas shift reaction 55 kJ/mol [5]. The effectiveness factor is in the range of 0.05- 0.65 [5]
Deactivation
Sulfur and chlorides act as catalyst poisons. As mentioned earlier, the feed is initially pretreated for sulfur and chloride removal. Another major source of deactivation is coke deposition on the catalyst. Formation of carbon occurs by decomposition of hydrocarbon and CO on the surface of catalysts
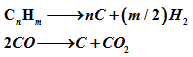
Gas phase non catalytic pyrolysis of hydrocarbons also contributes to coke deposition. All of these processes are favored at high temperature. High temperature also results in sintering, growth of metal crystallites and collapse of catalysts support which can cause decrease in activity.
Carbon can be removed by steam gasification. Carbon deposition can be avoided by operating at sufficiently high H2O to C ratio.
Secondary steam reforming
The exit gas from the primary reformer contains about 10-13 % CH4 which is further reformed in secondary reformer. Inlet temperature is 8000C and the outlet temperature is 900-10000C. These high temperatures drive the forward reaction and reduce the CH4 content to less than 1%. Outlet CO concentration is 10-13 %. Reactor is lined with ceramic material to prevent damage.
Catalysts
Catalyst must be able to withstand the severe high temperature. Typical catalysts are Ni supported on alumina or CaO substituted alumina. Potassium is not added due to volatility at this high temperature. Total BET surface area and surface area of active nickel for secondary reforming catalysts are smaller than that of primary reforming catalysts. This is because they are initially calcined at high temperature so that they are thermally stable at process conditions.