Effect of external transport on selectivity
For multiple reactions, obtaining high selectivity of the desired products is important. The effect of external transport on selectivity depends on the kinetics of the system. Selectivity at any location in a reactor can be defined as the ratio of the rates of the desirable to the undesirable product. The effect will be discussed at isothermal conditions only.
Case 1 : Isothermal-consecutive reaction:
Consider a first order irreversible consecutive reaction
![]()
The selectivity of B w.r.t C, SB can be written as ![]() (10)
(10)
The surface concentration can be expressed in term of bulk values.
For A,
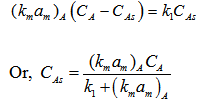
Similarly for B, (here B is diffused from surface to bulk)
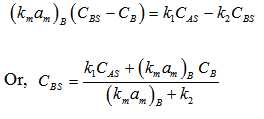
Here CA and CB are bulk concentrations of A and B, respectively
Substituting value of C AS
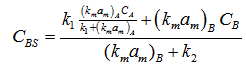
Substituting value of CAS & CBS in SB (10), selectivity can be obtained in term of bulk concentration
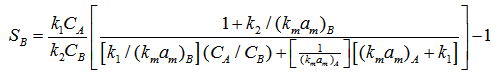 (11)
(11)
In absence of mass transfer resistances ![]() for both A & B
for both A & B
Then, the above equation reduces to
| (12) |
The term in brackets of equation (11) is less than unity hence comparison of (11) & (12) suggests that selectivity is reduced by mass transport effect. As mass transfer resistance becomes insignificant, the term in bracket in (11) approaches unity and it is in agreement with equation (12).
Qualitatively, the effect of external mass transfer can be explained as follows. The mass transfer limitation will reduce the surface concentration of A below the bulk value. This will reduce rate of formation of B by the first reaction. Again, due to mass transfer resistances in removal of B from the surface, concentration of B will increase near the catalyst surface and thereby result in an increase in the rate of disappearance of B to C by the second reaction. Thus, qualitatively it can be seen that formation of B is reduced while disappearance of B is increased due to external mass transfer effect. Consequently, selectivity of B with respect to C is reduced by external mass transfer limitations.
Case 2 : Parallel reaction (isothermal)
Consider two parallel first-order reactions from the same reactant with B as the desired product.
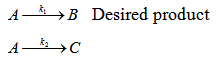
The selectivity of B w.r.t C is given by:
![]()
Hence selectively is independent of concentration. Hence, external mass transfer does not affect the selectivity in this case. For this type of parallel reaction, the rate is reduced by mass transfer but the selectivity is unchanged.
If the parallel reactions are completely independent
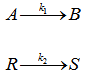
Selectivity is then given as
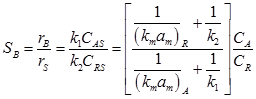
In absence of external resistance, selectivity is 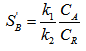
Effect of external mass transport on selectivity is given by ratio
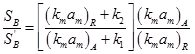
The two mass transfer coefficients are nearly the same for most reaction systems. Hence the ratio becomes
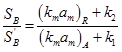
The selectivity in this case will decrease by external mass transfer only if ![]() For low mass transfer resistance, no effect on selectivity will be observed as
For low mass transfer resistance, no effect on selectivity will be observed as ![]() then
then ![]()
Book Reference :
• J. M. Smith, Chemical Engineering Kinetics, McGrawHill Book Company, 1981
• H. S. Fogler, Elements of Chemical reaction engineering, Prentice Hall of India., 1999
• J.J. Carberry, Chemical and catalytic reaction Engineering, Dover Publications, 2001
• O. Levenspiel, Chemical reaction engineering, John Wiley & sons, 1995