Effect of external mass transfer resistance on order of reaction
Consider a irreversible and gas phase reaction A → B. Assume true order of the reaction to be n = 2 . Hence actual reaction rate can be written as,
![]()
Now if the rate is mass transfer controlled, then from (6) rate is given as ![]()
Experimental data plotted as rate vs Cb will give a straight line (passing through origin). Now if the external diffusion is not taken into account then based on the experimental data it will be assumed that reaction is first order with respect to concentration because for first order reaction, ![]() , the plot of rate versus concentration gives a straight line with slope k passing through the origin. Hence in this case, the rate will be interpreted as
, the plot of rate versus concentration gives a straight line with slope k passing through the origin. Hence in this case, the rate will be interpreted as ![]() and the order will be interpreted as one. However, this interpretation will be a false conclusion as actual order of the reaction is two and it is appearing one only because external diffusion is controlling the process. Regardless of the intrinsic kinetics, if external mass transfer is controlling, the experimentally determined order will always appear as unity.
and the order will be interpreted as one. However, this interpretation will be a false conclusion as actual order of the reaction is two and it is appearing one only because external diffusion is controlling the process. Regardless of the intrinsic kinetics, if external mass transfer is controlling, the experimentally determined order will always appear as unity.
Effect of external mass transfer resistance on activation energy of reaction
Consider an irreversible and gas phase reaction A → B. Assume the reaction to be first order with respect to A.
Then, ![]() ............................................where k0= overall rate constant
............................................where k0= overall rate constant
Rates are measured over a nonporous catalyst at different temperature and observed rates are used to calculate the overall rate constant k0. The corresponding activation energy can be obtained from Arrhenius equation as follows
![]()
Here the activation energy E' is known as apparent activation energy and A' as apparent frequency factor. This E' is not the true activation energy corresponding to surface reaction as we will see in following section.
Now k0 overall rate constant is given as
Or , ![]() ------------------------------------------------ (8)
------------------------------------------------ (8)
If E is the true activation energy of the surface reaction then by substituting ![]() in equation (8)
in equation (8)
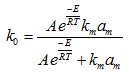 ...........................................................(9)
...........................................................(9)
(1) At high temperature , ![]()
Then from equation (9) ,![]() which is nearly a constant value as k m is relatively insensitive to temperature
which is nearly a constant value as k m is relatively insensitive to temperature
(2) At low temperature, ![]() , then
, then ![]() and a straight line is obtained on Arrhenius plot.
and a straight line is obtained on Arrhenius plot.
If the Arrhenius equation is plotted over the entire range of temperature, then at low temperatures, the slope corresponds to the correct (or true) activation energy E of the surface reaction. As temperature increases, a curve is obtained, which flattens to a horizontal line corresponding to constant value of ‘![]() '. Hence when experimental rate date for fluid solid catalytic reaction shows a curved Arrhenius plot then it is possible that mass transfer resistance is significant and external diffusion plays important role in kinetics. This situation is for isothermal conditions.
'. Hence when experimental rate date for fluid solid catalytic reaction shows a curved Arrhenius plot then it is possible that mass transfer resistance is significant and external diffusion plays important role in kinetics. This situation is for isothermal conditions.
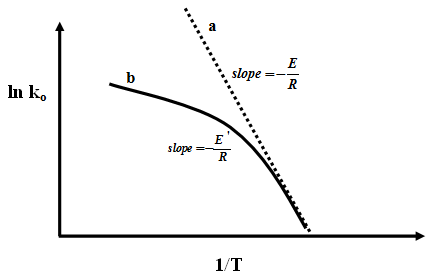 |
Fig. 2. Arrhenius plot for reaction (a) without external mass transfer resistance and (b) with external mass transfer resistance