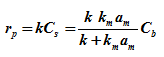Reaction rate in presence of significant external mass transfer limitation
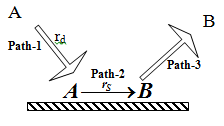
Consider a gaseous irreversible reaction A → B that is occurring on a solid catalyst. Assume the reaction to be of first order with respect to A. Since the reaction is irreversible, the concentration of B at the catalyst surface does not influence the rate. Also assume desorption of B to be very fast (path -3).
Hence the reaction is controlled by the following two steps
- (1) transportation of A from bulk to catalyst surface (path -1)
..........(2) reaction of A to B at catalyst surface (path -2)
Diffusion rate of A from bulk gas to surface is ![]() ------------ (1)
------------ (1)
Surface reaction rate is ![]() ----------------------- ----------- ----------- ---- (2)
----------------------- ----------- ----------- ---- (2)
Here rate is mol /s. g catalyst.
Where,

At steady state, rate of diffusion and surface reaction rate will be equal. Hence ![]()
And overall reaction rate can be expressed by either of two
![]() --------------------------------------------------- (3)
--------------------------------------------------- (3)
Here, ![]() is the steady state rate per unit mass of catalyst pellets.
is the steady state rate per unit mass of catalyst pellets.
From (1) , (2) and (3)
Or, Or , |
(4) |
Substituting the value of Cs in ![]()
Or |
(5) |
Where 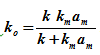 . The ko is called the overall rate constant
. The ko is called the overall rate constant