1. pI value and Net charge- The information of a pI will be allow you to calculate the net charge at a particular pH on a protein. As discussed above, a cation exchange chromatography can be use below the pI where as an anion exchange chromatography can be use above the pI value.
2. Structural stability-3-D structure of a protein is maintained by electrostatic and vander waal interaction between charged amino acid, Π-Π interaction between hydrophobic side chain of amino acids. As a result, protein structure is stable in a narrow range around its pI and a large deviation from it may affect its 3-D structure.
3. Enzymatic activity-Similar to structural stability, enzymes are active in a narrow range of pH and this range should be consider for choosing an ion-exchange chromatography.
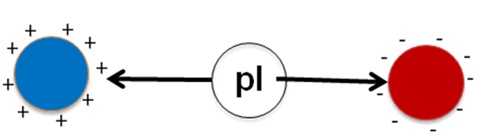
Figure 30.3: Change of charge with respect to the pI.
Operation of the technique-Several parameters needs to be consider to perform ion-exchange chromatography (Figure 30.4).
1. Column material and stationary phase-Column material should be chemically inert to avoid destruction of biological sample. It should allow free low of liquid with minimum clogging. It should be capable to withstand the back pressure and it should not compress or expand during the operation.
2. Mobile Phase-The ionic strength and pH are the crucial parameters to influence the property of the mobile phase.