-
Binding of ligands to proteins: Binding of a ligand to a protein usually does not affect the secondary structural elements significantly. However, such a binding can cause changes in the local tertiary structure. Binding of ligands accompanying such conformational changes can be studied using tertiary CD if the binding region happens to have one or more aromatic residues . Short peptides, on the other hand, can undergo large scale structural changes sometime involving completely switching from one secondary structure to another. Such changes can easily be observed using far UV CD.
- DNA structure: CD in the 200 – 300 nm region can be used to identify which structural isoform of DNA is present in the given sample. The left-handed helical DNA form, the Z-DNA was indeed identified using CD spectroscopy. The typical CD signatures of the B, Z, and A form of DNA are:
B-DNA: In its most common form i.e. B-DNA with ~10.4 bases per turn, a positive band ~275 nm, a crossover ~258 nm, and a negative band at ~240 nm are observed.
Z-DNA: A negative band ~290 nm and a positive band ~260 nm; a crossover between 180-185 nm.
A-DNA: A positive band ~260 nm, a negative band ~210 nm.
- Protein folding/unfolding: CD is used for studying the folding and unfolding of proteins. For monitoring the fast reactions such as protein folding, a single wavelength CD is recorded in a stopped flow experiment wherein the protein solution is mixed with a denaturant and CD is recorded as a function of time. Modern instruments take ~1 millisecond time between mixing and recording data allowing the understanding of the folding/unfolding events that occur on milliseconds to seconds timescale. A diagrammatic unfolding experiment is shown in figure 9.4
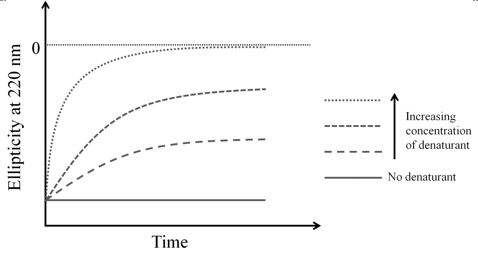
Figure 9.4 A diagram showing the kinetics of unfolding of a hypothetical protein. The protein is unfolded with different concentrations of a denaturant. Protein and denaturant are mixed in a stopped flow apparatus (mixing time typically ~1 ms) and changes in ellipticity are monitored over time.