CD of biomolecules
Most biomolecules are chiral and the biomacromolecules are composed of chiral components. Folding of biomacromolecules into higher order structures further imparts them the asymmetry. CD has not been used as much to study other biomolecules probably, as it has been used to study proteins.
CD of proteins
Proteins are usually composed of 20 amino acids, 19 of which (except glycine) are chiral. This chirality also reflects in the higher order structures that the polypeptides adopt; α-helix, for example, is a right handed helix. If a polypeptide adopting α-helical structure is synthesized using D-amino acids, it folds into the left-handed α-helix under identical conditions. The other structural features of a polypeptide backbone include β-sheets, that are comprised of extended polypeptide chains ; β-turns, that usually, but not essentially, link the β-strands in an antiparallel β-sheet; and unordered conformation. CD spectra of the proteins contain information about the asymmetric features of the polypeptide backbone. Furthermore, it can provide information about the orientation of the side chains. CD, therefore, is capable of providing information about the structure of proteins which in turn helps understanding their function. The chromophore that provides information about the conformation of the peptide backbone is the peptide bond (Figure 9.1); the spectra are therefore recorded in the far UV region, the region where peptide bond absorbs.
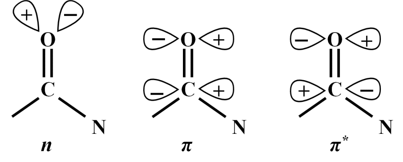
Figure 9.1 The peptide bond showing molecular orbitals involved in electronic transitions |