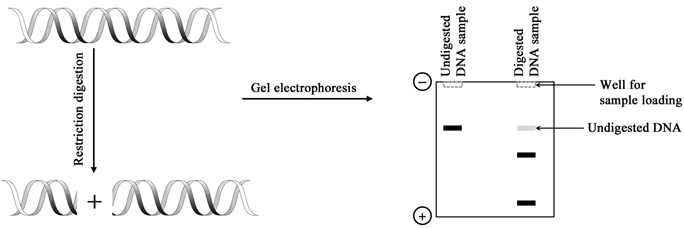
Figure 2.1 Restriction digestion of a DNA molecule assessed by agarose gel electrophoresis
Quantification of an analyte, as has been discussed in the previous lecture, is among the most common applications of analytical tools. You may be familiar with the use of UV/visible light for recording absorption of organic molecules to determine the concentration of the compound. It is therefore clear that light or electromagnetic radiation can interact with the matter providing useful information about it. Interaction of electromagnetic radiation with matter is termed as spectroscopy. Absorption of UV/visible radiation is associated with electronic transitions in the molecules; UV/Visible spectroscopy is therefore also referred to as the electronic spectroscopy. Absorption of ultraviolet and/or visible radiation is the most commonly employed method to estimate the concentration of biomolecules such as proteins, peptides, nucleotides, nucleic acids, carbohydrates, and lipids. Absorption at 260 nm and 280 nm provides information about the nucleic acid contamination in protein preparations. Phenol is commonly used to isolate nucleic acids; ![]() is used to determine phenol contamination in nucleic acid preparations and has become a routinely used method in molecular biology laboratories. Electronic spectroscopy goes beyond quantification of biomolecules: fluorescence spectroscopy is used to study various biological processes viz. protein folding/unfolding, binding studies, etc. Electronic circular dichroism spectroscopy is a chiroptical method and finds applications in analyzing protein and peptide structures, protein folding/unfolding, binding studies, etc.
is used to determine phenol contamination in nucleic acid preparations and has become a routinely used method in molecular biology laboratories. Electronic spectroscopy goes beyond quantification of biomolecules: fluorescence spectroscopy is used to study various biological processes viz. protein folding/unfolding, binding studies, etc. Electronic circular dichroism spectroscopy is a chiroptical method and finds applications in analyzing protein and peptide structures, protein folding/unfolding, binding studies, etc.
Infrared spectroscopy probes the vibrational frequencies in the molecules; the frequency of vibration depends on the strength of the bond and the atoms involved thereby allowing identification of functional groups present in the organic molecules. As the absorption depends on the concentration, infrared spectroscopy can also be utilized for determining the concentrations of the analytes. The vibrational frequencies of the bonds are sensitive to the conformation of the molecule as well as the interactions of the atoms involved. Infrared spectroscopy can therefore provide information about the conformations of the molecules. In fact, infrared spectroscopy is often used to determine the secondary structures of the polypeptides. Advent of nuclear magnetic resonance (NMR) spectroscopy in 1940s revolutionized the analysis of small molecules. When used alongside infrared spectroscopy, NMR spectroscopy can quickly provide the complete structure of the molecules. Advancement in the hardware and development of the experimental methods has made NMR spectroscopy one of the most powerful weapons in a chemist's and biochemist's arsenal. NMR is routinely employed to study the structure and dynamics of biomacromolecules. In fact, NMR is the only tool that provides atomic resolution structure of the molecules in solution. This is a big plus for NMR spectroscopy over X-ray crystallography that needs a crystal for determining the atomic resolution structure. Furthermore, solid state NMR spectroscopy can be used to study the solid samples including single crystals. Atomic resolution structure determination requires the biomolecules with very high purity. High purity biological macromolecules are obtained through one or more chromatographic methods. The principle underlying the separation of molecules is their partitioning between a stationary and a mobile phase. The partition coefficient of a molecule depends on its physicochemical properties and molecules can be separated based on their size, charge, hydrophobicity or affinity to a particular ligand. Chromatographic techniques can also provide analytical information, e.g. molecular weight can be determined using size exclusion chromatography wherein there is a relationship between the molecular weight and the elution volume. NMR spectroscopy, however, has come a long way since its discovery and it is now possible to determine the structures of biomolecules in their native milieu i.e. inside the living cells.