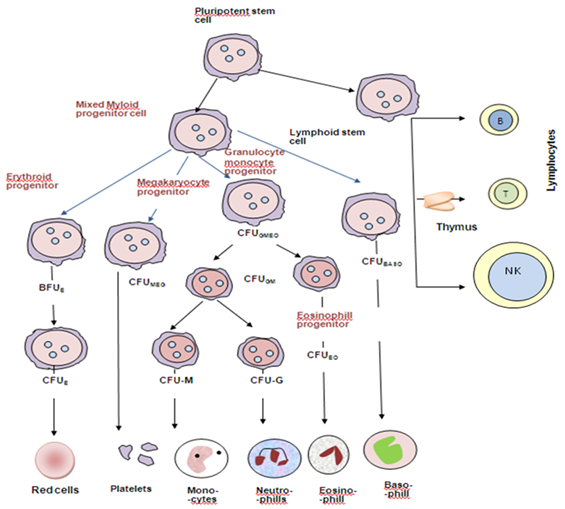
Figure 1: Diagrammatic representation of the bone marrow pluripotent stem cell and the cell lines that arise from it. Various progenitor cells can be identified by culture in semi-solid medium by the type of colony they form. Baso, basophil; BFU, burst-forming unit; CFU, colony-forming unit; E, erythroid; Eo, eosinophil; GM, granulocyte, monocyte; Meg, megakaryocyte; NK, natural killer cell (Hoffbrand et al. 2011).
Table 2 Growth factors in haemopoiesis
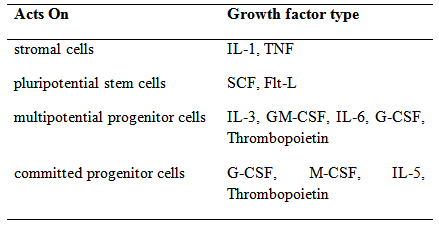
Legend: Flt-L, Flt ligand; G- and GM-CSF, granulocyte and granulocyte–macrophage colony-stimulating factor; IL, interleukin; M-CSF, macrophage colony-stimulating factor; SCF, stem cell factor; TNF, tumour necrosis factor.
Growth factor receptors and signal transduction
The biological effects of growth factors are mediated through specific receptors on target progenitor cells. Receptors like granu locyte macrophage colony-stimulating factor GMCSF-R are from the haematopoietin receptor superfamily which possesses the capacity to dimerize after binding their ligand. This results in cascade of intracellular signal transduction pathways of which the three major ones are the Janus associated kinase or JAK/STAT, the mitogen activated protein (MAP) kinase and the phosphatidylinositol 3 (PI3) kinase pathways (see Figure 2). The JAK proteins are tyrosine-specific protein kinases that associate with the intracellular domains of the growth factor receptors. A growth factor molecule binds simultaneously to the extracellular domains of two or three receptor molecules, resulting in their aggregation. JAKs then phosphorylate members of the signal STAT family of transcription factors resulting in their dimerization and translocation from the cell cytoplasm across the nuclear membrane to the cell nucleus where specific genes are transcribed. JAK also activates the MAPK pathway which is in turn controlled by Ras. Different domains of the intracellular receptor protein may signal for the different processes (e.g. proliferation or suppression of apoptosis) mediated by growth factors. Other growth factors like SCF, Flt-3L and macrophage colony-stimulating factor (M-CSF) bind to receptors that have an extracellular immunoglobulin-like domain linked. Growth factor binding results in dimerization of these receptors and consequent activation of the tyrosine kinase domain. Phosphorylation of tyrosine residues in the receptor itself generates binding sites for signalling proteins which initiate complex cascades of biochemical events resulting in changes in gene expression, cell proliferation and prevention of apoptosis.