Adhesion proteins of Matrix
Matrix adhesion proteins are accountable for connecting the components of the matrix to one another and to the surfaces of cells. They act together with collagen and proteoglycans to direct matrix organization and bind to integrins. The first of its kind is fibronectin, which is the main adhesion protein of connective tissues. Fibronectin is a glycoprotein with two polypeptide chains, of 2500 amino acids. Additionally fibronectin possess binding sites for both collagen and GAGs thus crosslinking these matrix. A specific site on the fibronectin molecule is responsible for recognizing cell surface receptors like integrins attaching of cells to the extracellular matrix. Prototype of adhesion proteins belong to the laminin family with the property of self assembly into mesh like networks.
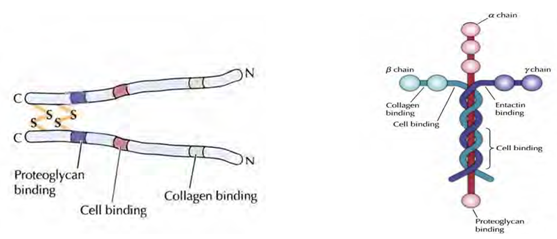
Figure 4: An illustration of matrix associated proteins. A. Fibronectin. B. Laminin
Cell matrix interaction
Cells remain attached to the extracellular matrix through the aid of cell surface receptors such as integrins. The integrins belong to the family of transmembrane proteins consisting of one α and one β subunits. The integrins bind to short amino acid sequences present in multiple components of the extracellular matrix, including collagen, fibronectin, and laminin. In addition to attaching cells to the extracellular matrix the integrins also provide anchors for the cytoskeleton resulting in stability of the cell matrix junctions. Integrins interact with the cytoskeleton at two junctions of the extracellular matrix known as the focal adhesions and hemidesmosomes. Focal adhesions attach a variety of cells, including fibroblasts, to the extracellular matrix and hemidesmosomes mediate epithelial cell attachments at with a specific integrin (Figure 5).