During the current lecture we shall discuss about Extracellular matrix and their role in cell signaling and adhesion
Extracellular matrix
Animal cells are surrounded by extracellular matrix beyond the immediate vicinity of their plasma membrane, filling spaces between cells and adhering cells together. Extracellular matrices are of various types consisting of secreted proteins and polysaccharides and are most abundant in connective tissues. One of the examples of extracellular matrix is the basal laminae. It is a continuous sheet of 50 to 200 nm thickness and on top of which a thin layer of epithelial cells rest. Such basal laminae surround muscle cells, adipose cells, and peripheral nerves. The differences between various types of extracellular matrices result from both quantitative variations in the types or amounts of these different constituents and from modifications in their organization. The three major components of extracellular matrix are matrix proteins, matrix polysaccharides and the matrix adhesion proteins. The major components of the extracellular matrix have been illustrated in Figure 1.
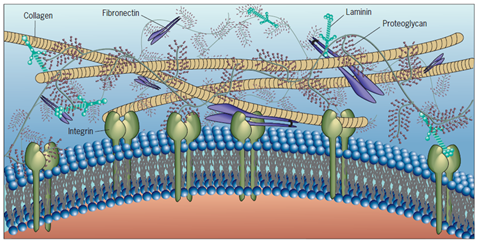
Figure 1: An overview of the extracellular matrix molecular organization. The proteins; fibronectin, collagen, and laminin contain binding sites for one another, as well as binding sites for receptors like integrins that are located at the cell surface. The proteoglycans are huge protein polysaccharide complexes that occupy much of the volume of the extracellular space. This figure has been adapted from Cell and Molecular Biology Concepts and Experiments by Karp, 2010.
Structural proteins of Matrix
Matrix proteins are fibrous in nature. The major structural protein is collagen whose secondary structure is a triple helix. The collagens belong to large family of proteins and are characterized by the formation of triple helices in which three polypeptide chains are wound tightly around one another in a ropelike manner. The different collagen polypeptides can assemble into 42 different trimers. The triple helix domains of the collagens consist of repeats of the amino acid sequence Gly-X-Y. The most abundant type is collagen type I and is one of the fibril forming collagens that are the basic structural components of connective tissues (Figure 2) . Elastin is another matrix protein, which gives elasticity to tissues, allowing them to stretch when needed and then return to their original state. They are present in blood vessels, the lungs, in skin, and the ligaments. Elastins are synthesized by fibroblasts and smooth muscle cells.