| |
Another problem with the classical theory is that it would predict a time lag between the time light falls on a surface and the instant photoelectrons are emitted. The reason why one would expect such a time lag is that the surface are of an atom is very small, as a result of which an atom can only absorb a small fraction of energy that falls on the surface. The following example gives a rough estimate of the expected time lag. However, it is observed that the emission of electrons is practically instantaneous, with time lag, if any, being less than 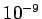 seconds. seconds. |