| |
The electrons which are more tightly bound to the metal (e.g. electrons which lie two or three atomic layers below the surface) require more energy to be removed. We define Work Function 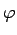 of a metal as the minimu energy that must be supplied to an electron at the metal surface to dislodge it from the metal. Such electrons are emitted with maximum possible kinetic energy. Thus Einstein's equation becomes of a metal as the minimu energy that must be supplied to an electron at the metal surface to dislodge it from the metal. Such electrons are emitted with maximum possible kinetic energy. Thus Einstein's equation becomes |
| |
Since kinetic energy cannot be negative, the above equation implies the existence of a minimum frequency 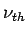 for photoemission to take place for photoemission to take place |
| |
Using this, we can reqrite
rewrite Einstein's equation as |
| |
To stop such maximum energy electrons from reaching the anode, we must apply a reverse potential 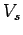 , given by , given by 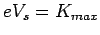 . Thus . Thus |