|
The maximum kinetic energy of the photoelectrons depend on the frequency of incident radiation and not on its |
|
The emission of photoelectrons is almost instantaneous. i.e. there is no time lag between the emission of |
|
|
electrons and switching on of the light source. |
 |
Failure of Classical Wave Theory
According to the classical wave theory, when electromagnetic wave falls on the surface of a metal, an atom on the surface will absorb energy from the electric field of the wave. The rate at which the energy is absorbed depends on the surface area of the atom. An electron can be dislodged from an atom once it absorbes sufficient amount of energy. By increasing the intensity of light (irrespective of its frequency) more energy can be transferred to the atom causing electrons to be ejected.
|
| |
What is observed is that unless 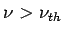 photoelectrons are not emitted, no matter how intense the radiation is. photoelectrons are not emitted, no matter how intense the radiation is.
Further, according to wave theory, the kinetic energy of emitted electrons would increase with the intensity of light as it would impart more energy to an electron. However, the kinetic energy of photoelectrons is found to depend only on the frequency of radiation and not on the intensity. |