 |
Planck's explanation of black body radiation was revolutionary as it suggested that atoms could exchange energy only in multiples of quantum of energy. Five years later, in 1905, Einstein put forward a theory of photoelectric effect which suggested that the quantum of energy was not a property associated with the radiation emitted by atoms but is a property of radiation itself. Radiation, according to Einstein's theory consists of discrete bundles of energy, called photons. Thus, electromagnetic energy is seen as a collection of photons. A photon is characterized by an energy 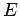 , related to the frequency by the relationship , related to the frequency by the relationship |
| |
Further, each photon carries a momentum 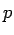 given by given by |
| |
In Einstein's special theory of relativity, the energy of a particle of rest mass  and momentum and momentum 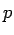 is given by is given by |
| |
which implies that photons have zero rest mass. |