| Mechanism 2 |
NO (g) + NO (g)
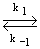 N2O2 N2O2 |
(28.28) |
| |
N2O2 (g) + O2 (g)
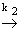 2NO2 (g) 2NO2 (g) |
(28.29) |
| In this mechanism a rapid equilibrium is not established in eq (28.28) but a steady state approximation can be used for the concentration of N2O2. |
| |
| d [ N2 O2 ] / dt = k 1 [NO]2 - k -1 [ N2 O2] - k2 [ N2O2] [O2] |
(28.30) |
| Setting this equal to zero we get, for the concentration of [N2O2], |
| |
| [N2O2] = k 1[NO]2 / (k-1 + k 2 [O2]) |
(28.31) |
| This equation for the constant (steady) value of [N2O2] is satisfied if k-1 is greater than k2 and k1. Only in such a situation is a small value of [ N2O2] is present, satisfying the SSA. In such a situation, [ N2O2] = (k1 / k-1) [NO]2 and the rate law for eq (28.29) becomes |
| |
|
| (1/2) d [ N O 2 ] / dt = k2 [ N2 O2 ] [ O2 ] = (k2 k 1/ k -1) [ NO ] 2 [ O2 ] |
(28.32) |
| Notice that both mechanisms give the same rate law! The only way to distinguish between the mechanisms is by detecting NO3 and / or N2O2. At present the second mechanism is favoured. |
| |