Even for reactions that appear very simple such as A
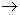 [P] where P refers to products, the mechanism can be fairly involved. This is because for reactions to occur, molecules need to be activated or initiated through collisions (unlike radioactivity which is spontaneous!). Collisions could be with other molecules M that are present in the reaction mixture, which are usually in large amounts. The reaction sequence is [P] where P refers to products, the mechanism can be fairly involved. This is because for reactions to occur, molecules need to be activated or initiated through collisions (unlike radioactivity which is spontaneous!). Collisions could be with other molecules M that are present in the reaction mixture, which are usually in large amounts. The reaction sequence is
|