The mechanism of a chemical reactions consist of the complete set of elementary reactions that contribute to the over all reaction process. Ideally, we need to know the rate constant for all these elementary reactions(both forward and reverse rate constant), but chemists are usually quite happy if they can unambiguously identify all the elementary reactions and the associated transitions state structures and geometrices. the transition state or activated complex is a state intermediate between reactions and products and with the current advances in spectroscopy it is possible to identify these transition states. In the present section, we will describe the mechanisms of a few reactions. |
|
| In understanding these mechanisms and using the rate constants of the elementary reactions in the over all rate constants certain approximations (or assumptions ) help greatly in simplifying the rate equations. We will first elaborate o these. |
|
| a) The steady state approximation. |
|
|
| In this approximation, it is assumed that the concentrations of the reactive intermediates remain constant and small during the major part of the reaction process. We will illustrate this approximation for the following consecutive reaction scheme. |
| |
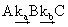 |
(28.2) |
| |
| An example for such a reaction is |
| |
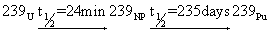 |
(28.3) |
| |
| Since each step here is an elementary reaction, rate laws can be written as follows, |
| |