In the newer and more accurate theory of matter given by Heisenberg, Schrödinger and Dirac, the description of matter is not based on the simultaneous specification of positions and momenta of particles. This implies that we cannot speak of the trajectories of these subatomic particles at all. Indeed, the Heisenberg uncertainty principle states that the position and the momentum of a particle cannot be determined simultaneously and the product of the uncertainty of the position 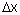 and the uncertainty of momentum and the uncertainty of momentum 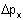 is given by is given by
|