- Excess carriers, essential for device operation, are created by
optical excitation, electron bombardment, or injected across a forward-biased
p-n junction.
- These excess carriers can dominate the conduction process in semiconductor materials.
- This includes photons in the optical range as well as those in the
infrared region.
- Photons of various wavelengths (frequencies) are directed at the
sample, and their relative transmission is measured.
- Note: photons having energies greater than the band gap energy are
absorbed (the sample behaves opaque for this kind of illumination),
whereas those having energies less than the band gap energy are transmitted
(the sample behaves transparent), this experiment gives an accurate
measure of the band gap energy.
- When photons having energies h Eg are absorbed, they create EHPs
and the probability of this absorption is very high, since there are
lots of electrons in the valence band and lots of empty states in
the conduction band.
- Electrons excited to EC may initially have energies much higher
than EC, however, they lose this excess energy due to scattering with
the lattice until their equilibrium energy becomes equal to EC.
- Note: these EHPs are called excess carriers, since they are out
of balance, and, thus, would eventually recombine.
- However, while these excess carriers remain in the respective bands, they can contribute to the current conduction.
- The transmitted intensity It of a beam of photons of wavelength
through a sample of thickness t can be given by
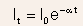
where is
called the absorption oefficient, and varies with materials and photon
wavelength
is
called the absorption oefficient, and varies with materials and photon
wavelength
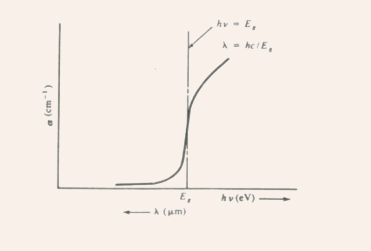
Fig.3.1 The variation of the absorption coefficient as a function
of the wavelength of the incident light.
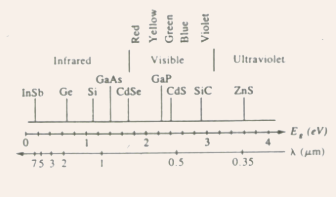
Fig.3.2 Band gaps of some common semiconductors relative to the optical
spectrum.
- Absorption cutoff occurs at
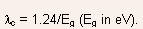
- GaAs, Si, Ge, and InSb band gaps are such that
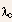 c occurs beyond the visible region (in the infrared), whereas GaP
and CdS have band gaps with
c occurs beyond the visible region (in the infrared), whereas GaP
and CdS have band gaps with 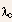 c falling within the visible range.
c falling within the visible range.
- When recombination occurs between a conduction band electron and a
valence band hole, the energy released can be given off in the form
of light (luminescence).
Direct band-to-band recombination in direct band gap semiconductors have a much higher probability of light emission as compared to those in indirect materials. - Broadly divided into three categories:
- Photoluminescence: if the recombining carriers were caused by optical excitation.
- Cathodoluminescence: if the recombining carriers were caused by high energy electron bombardment.
- Electroluminescence: if the recombining carriers were caused by injection of excess carriers (by forward biasing a p-n junction, for example).
- For steady state excitation, the recombination rate and the generation rate for EHPs are equal, and one photon is emitted for each photon absorbed.
- Direct band-to-band recombination is a fast process with typical
lifetime of excess carriers 10 8 sec => known as fluorescence (example:
fluorescent lamp).
- In some indirect materials, the trap states within the band gap
captures carriers, and slows down the recombination process, thus,
emission continues for seconds or minutes after the excitation is
removed => known as phosphorescence and the materials are known
as phosphors.
- The trap states can hold the carriers for indefinite times, and the carriers can either get reexcited to the conduction band or fall to the valence band (and, thus, recombine) => this creates the delay between excitation and recombination.
EXAMPLE 3.1: A 0.5 m thick sample of
(a) Find the total energy absorbed by the sample per second (J/sec).
(b) Find the rate of excess thermal energy given up by the electrons to the lattice before recombination (J/sec).
(c) Find the number of photons per second given off from recombination events, assuming perfect quantum efficiency.
SOLUTION:
- (a)The transmitted intensity
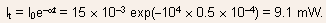
Therefore, the absorbed power
(15 9.1) mW = 5.9 mW = 5.9 10 3 J/sec.
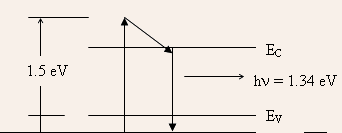
- (b)Since the energy of the incident photon is greater than the
band gap energy, hence, the excess energy of the excited electron
will be dissipated as heat to the lattice. The fraction of energy
converted to heat is given by
(1.5 1.34)/1.5 = 0.107. Thus, the amount of energy converted to heat per second
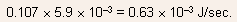
- (c) For perfect quantum efficiency, one photon is emitted for each
photon absorbed. Thus, the number of photons emitter per second

or, alternately, recombination radiation accounts for 5.9 0.63 = 5.27 mW at 1.34 eV/photon. Thus,
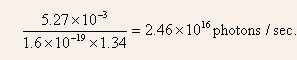
- Best example: cathode ray tube (CRT) basis of television sets, oscilloscopes,
and other display systems.
- Electrons emitted from the heated cathode are accelerated towards
the anode by high field, deflected by electric or magnetic fields by
the horizontal and vertical plates, and made to hit the screen (coated
with a phosphor) at desired locations.
- When electrons hit these phosphors, the energy of the electrons gets
transferred to the electrons of the phosphors, and they get excited
to higher states, and eventually fall to the ground state, thus causing
recombination and light emission.
- Three phosphor dots are used for each pixel, capable of transmitting three primary colors: red, green, and blue (RGB) thus by varying the intensity and position of the electron beam, a wide range of colors and picture can be attained.
- Best examples: LEDs and LASERs, where carriers are injected across a forward biased p-n junction and are made to recombine (either naturally or by carrier confinement) => called injection electroluminescence.
- Photoconductivity: increase in the conductivity of a sample due to
excess carriers created by optical excitation.
- With excitation turned off, the photoconductivity decreases to zero since all excess carriers eventually recombine.
- Direct recombination occurs spontaneously, i.e., the probability
that an electron and a hole will recombine is constant in time, which
leads to an exponential solution for the decay of the excess carriers.
- The net rate of change in the conduction band electron concentration
at any time t
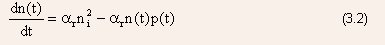
where the first term is the generation rate and the second term is the recombination rate.
- Let excess EHPs n and p (with n = p, since they are created in
pairs) are created at t = 0 by a short flash of light.
- Define n(t) and p(t) (again n = p) as the instantaneous excess carrier
concentrations and n and p for their values at t = 0.
- Note: n(t) = n0 + n(t), and p(t) = p0 + p(t).
- Thus,
- Similarly, excess holes in an n-type material recombine with a
rate
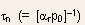
- Note: for direct recombination, the excess majority carriers (which
is equal to the excess minority carriers) decay at exactly the same
rate as the minority carriers, however, there is a large percentage
change in the minority carriers as compared to the majority carriers.
- A more general expression for carrier lifetime for near not sufficiently
extrinsic samples is
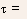

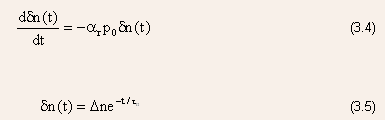
where