Lz would take any value from +L to –L and, therefore, when the magnetic field is turned on, different atoms will move up and down by different amounts till they strike the collecting plate. The result would be a spreading out of the traces as shown in Figure 2.2(b). However, Stern and Gerlach found that this was not the case. On the other hand, the beam split into a few sub-beams, each of which made a well-defined separate trace on the plate. The number of traces depends on the type of atoms forming the beam. This is possible only if the angular momentum is quantized. When L is quantized, then the only possible values of Lz are given by mlh where the magnetic quantum number ml can take (2l +1) values ranging from –l to +l , l being the orbital quantum number. Thus the beam must be split into (2l +1) sub-beams. Since (2l +1) is odd, it would imply that the number of traces on the plate would always be odd. However, this was not observed in the results. For many atoms (silver, hydrogen, lithium, sodium, potassium, copper and gold), it was found that the beam splits into two components, an even number. A clear explanation came only after the hypothesis of electron spin was proposed in 1925 because the spin would also contribute to the magnetic moment of an electron.
In case of silver, the valancy is one. It is now known that the orbital and spin magnetic moments of all but the valance electron in a silver atom cancel out. Hence, the orbital angular momentum, and the orbital magnetic moment is zero. Therefore, if spin were not there, a silver beam would not be split when the magnetic field is on. Now, if spin is taken into account, it can take up only two orientations in a magnetic field, corresponding to ms = ± 1/2. Hence, there would be two sub-beams, which are actually observed. Therefore, the Stern-Gerlach experiment was a direct confirmation of space quantization and the concept of electron spin.
The Quantum Mechanics of Spin:
New quantum theory based on Heisenberg's discovery of matrix mechanics and Schrödinger's discovery of wave mechanics not only predict quantization of energy and provide a prescription to determine the energy difference between the levels, but also allows one to calculate the probability of transition between different quantized energy states. In wave mechanics, the Schrödinger equation for a single particle tells us how the wavefunction evolves in time and space. It canbe expressed as
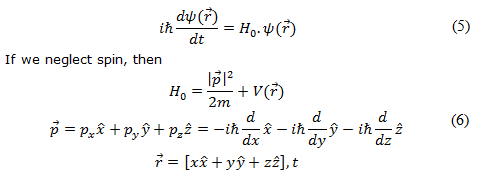
where ψ(r) is the wavefunction. The equation with 'hats' are unit vectors along the coordinate axes. Note that eqn.(5) does not include the spin part and hence the question raises here is how to include the 'spin' part?