Nuclear Magnetic Resonance Spectroscopy:
Nuclear Shielding:
The power of NMR is based on the concept of nuclear shielding. The magnetic field at the nucleus is not equal to the applied magnetic field. This is because, every atom is surrounded by electrons, which orbit the nucleus. When charged particles move in a loop, they create a magnetic field. Hence, the field produced by the movement of electron is felt by the nucleus. Therefore, the field experienced by the nucleus is not same as applied field, known as shielding. Therefore, the change in the energy levels requires a different frequency to excite the spin flip. The shielding allows for structural determination of molecules.
Consider the s-electrons in a molecule as displayed in Figure 35.05. They have spherical symmetry and circulate in the applied field, producing a magnetic field which opposes the applied field. This means that the applied field strength must be increased in order for the nucleus to absorb at its transition frequency. This up-field shift is also termed diamagnetic shift.
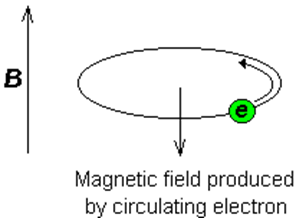
Figure 35.05: Schematic of nuclear shielding.
Similarly, electrons in p-orbitals having no spherical symmetry produce comparatively large magnetic fields at the nucleus giving a low-field shift and termed as a paramagnetic shift.