Nuclear Magnetic Resonance Spectroscopy:
NMR Spectrum:
Figure 35.08 displays another NMR spectrum of CH3Br. Since CH3Br contains only a single type of hydrogen atom, only a single peak at 2.7 ppm was observed.
The position is slightly de-shielded due to the presence of the Br.
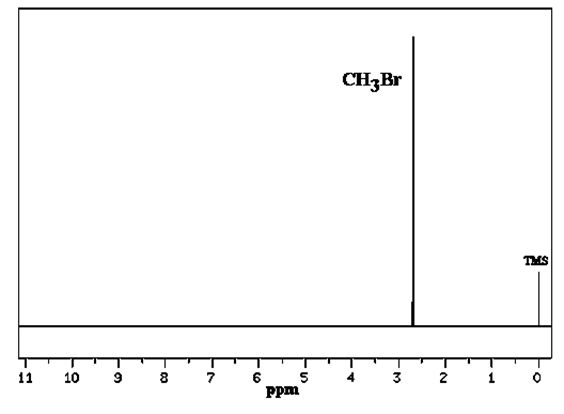
Figure 35.08: NMR Spectrum of methyl bromide.
Quiz 35:
(Q35.1) What are the rules exist to determine the net spin of a nucleus?
(Q35.2) Why do we need the magnetic field in NMR spectroscopy?
(Q35.3) What is called nuclear shielding?
(Q35.4) Describe the chemical shift?
(Q35.5) How does the NMR spectrum provide on the property of the materials?