LEED: Principle of operation:
By the principles of wave-particle duality, the beam of electrons may be equally regarded as a succession of electron waves incident normally on the sample. These waves will be scattered by regions of high localized electron density, i.e. the surface atoms, which can therefore be considered to act as point scatterers.
The wavelength of the electrons is given by the de-Broglie relation:
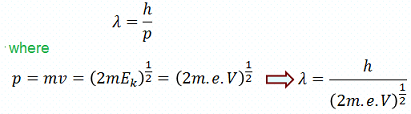
m is mass of electron, v is velocity, e is electronic charge and V is acceleration voltage in eV.