![]() and
and ![]() diagrams:
diagrams: ![]() and
and ![]() diagrams are shown in Fig.2.6. From these diagrams as well as from Fig.3, it can be seen that there is a large difference in densities in the liquid and gas phases of a fluid at low temperature. In the magnetic system, there is a large difference in spontaneous magnetization below
diagrams are shown in Fig.2.6. From these diagrams as well as from Fig.3, it can be seen that there is a large difference in densities in the liquid and gas phases of a fluid at low temperature. In the magnetic system, there is a large difference in spontaneous magnetization below ![]() . As
. As ![]() is approached from below, the density difference
is approached from below, the density difference ![]() of a fluid system and the spontaneous magnetization M of a magnetic system tend to zero. A quantity which is non-zero below
of a fluid system and the spontaneous magnetization M of a magnetic system tend to zero. A quantity which is non-zero below ![]() and zero above
and zero above ![]() is called the order parameter of the transition. Thus,
is called the order parameter of the transition. Thus, ![]() and M serve as order parameter of the fluid and magnetic system respectively. Note that below
and M serve as order parameter of the fluid and magnetic system respectively. Note that below ![]() the order parameter is multivalued where as it is single valued (zero) above
the order parameter is multivalued where as it is single valued (zero) above ![]() . Thus, the order parameter has a branch point singularity at
. Thus, the order parameter has a branch point singularity at ![]() .
.
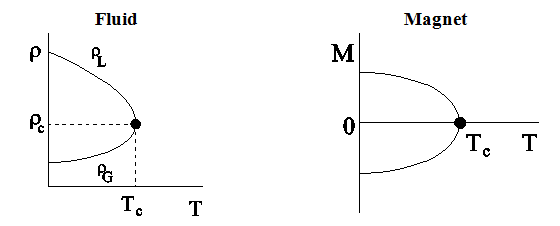
Figure 2.6: Schematic plot of density ρ against temperature T for fluid system and of spontaneous magnetization M against temperature T for magnetic system. |
The critical point ![]() at which the transition occurs is found to be dependent on the details of interatomic interactions or underlying lattice structure and varies from material to material. Critical temperatures of different materials are listed in table 2.1.
at which the transition occurs is found to be dependent on the details of interatomic interactions or underlying lattice structure and varies from material to material. Critical temperatures of different materials are listed in table 2.1.
Fluids |
|
|
|
Magnets |
|
Water |
647.5 |
218.50 |
0.325 |
Fe |
1043.00 |
Alcohol |
516.6 |
63.10 |
0.280 |
Ni |
627.20 |
CO2 |
304.2 |
72.80 |
0.460 |
CrBr3 |
32.56 |
Argon |
150.8 |
48.34 |
0.530 |
EuS |
16.50 |
Table 2.1: List of critical parameters for different fluid and magnetic systems. For magnetic systems, other critical parameters are spontaneous magnetization (M) and external magnetic field (H). Both M and H are zero at the critical point. Data have been collected from reference [3].