4. Continuous Phase transition or Critical phenomena:
Critical phenomena are the characteristic features that accompany the second order phase transition at a critical point. The critical point is reached by tuning thermodynamic parameters (for example temperature T or pressure P or both). A critical phenomenon is seen as T(P) approaches the critical point ![]() . In order to understand the characteristic features appearing at the critical point, one must study the macroscopic properties of the system at the critical point. In principle, all macroscopic properties can be obtained from the free energy or the partition function of a given system. However, since the critical phenomena, a second order phase transition or a continuous phase transitions, involve discontinuities in the response functions (which are second derivatives of the free energy function) at the critical point there must be singularities in the free energy at the critical point. On the other hand, the canonical partition function of a finite number of particles is always analytic. The critical phenomena then can only be associated with infinitely many particles, i.e. in the "thermodynamic limit'', and to their cooperative behaviour. The study of critical phenomena is thus essentially related to finding the origin of various singularities in the free energy and characterizing them.
. In order to understand the characteristic features appearing at the critical point, one must study the macroscopic properties of the system at the critical point. In principle, all macroscopic properties can be obtained from the free energy or the partition function of a given system. However, since the critical phenomena, a second order phase transition or a continuous phase transitions, involve discontinuities in the response functions (which are second derivatives of the free energy function) at the critical point there must be singularities in the free energy at the critical point. On the other hand, the canonical partition function of a finite number of particles is always analytic. The critical phenomena then can only be associated with infinitely many particles, i.e. in the "thermodynamic limit'', and to their cooperative behaviour. The study of critical phenomena is thus essentially related to finding the origin of various singularities in the free energy and characterizing them.
Let us consider more carefully the two classic examples of second order phase transition involving condensation of gas into liquid and transformation of paramagnet to ferromagnet. In the liquid-gas or fluid system the thermodynamic parameters are ![]() and in magnetic system the corresponding thermodynamic parameters are
and in magnetic system the corresponding thermodynamic parameters are ![]() . One may note the correspondence between the thermodynamic parameters of fluid and magnetic systems as:
. One may note the correspondence between the thermodynamic parameters of fluid and magnetic systems as: ![]() and
and ![]() . In the case of fluid, instead of volume V we will be considering the density ρ as a parameter. The equations of states in these systems are then given by
. In the case of fluid, instead of volume V we will be considering the density ρ as a parameter. The equations of states in these systems are then given by ![]() and
and ![]() respectively. A second order phase transition is a qualitative change in the system behaviour at a sharply defined parameter value, the critical point, when the parameter changes continuously. The critical points are usually denoted by
respectively. A second order phase transition is a qualitative change in the system behaviour at a sharply defined parameter value, the critical point, when the parameter changes continuously. The critical points are usually denoted by ![]() and
and ![]() . Commonly, phase transitions are studied varying the temperature T of the system and a phase transition occurs at
. Commonly, phase transitions are studied varying the temperature T of the system and a phase transition occurs at ![]() . We will be describing the features at the critical point by considering different phase diagrams such as
. We will be describing the features at the critical point by considering different phase diagrams such as ![]() ,
, ![]() ;
; ![]() ,
, ![]() ;
; ![]() ,
, ![]() of the full three dimensional phase space of
of the full three dimensional phase space of ![]() or
or ![]() .
.
![]() and
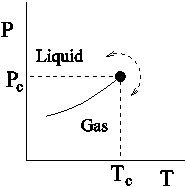
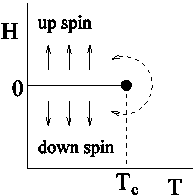
and ![]() diagrams: In Fig.2.4,
diagrams: In Fig.2.4, ![]() and
and ![]() diagrams are shown. It can be seen that the first order transition line (the vapour pressure curve for the fluid system and
diagrams are shown. It can be seen that the first order transition line (the vapour pressure curve for the fluid system and ![]() line for the magnetic system) terminates in a critical point at
line for the magnetic system) terminates in a critical point at ![]() . This means that the liquid can be converted to gas continuously without crossing the first order transition line following a path shown by a curved dotted line. Similarly, in the case of magnetic system a continuous change from up spin region to down spin region is also possible.
. This means that the liquid can be converted to gas continuously without crossing the first order transition line following a path shown by a curved dotted line. Similarly, in the case of magnetic system a continuous change from up spin region to down spin region is also possible.
Fluid |
Magnet |
|
|
Figure 2.4: Schematic plot of pressure P versus temperature T for a fluid in a gas-liquid transition and of magnetic field (H) versus temperature T for an Ising ferromagnet. The solid line is the first order transition line which ends at a critical point at Tc. |
|